Modular Design of High-Brightness pH-Activatable Near-Infrared BODIPY Probes for Noninvasive Fluorescence Detection of Deep-Seated Early Breast Cancer Bone Metastasis: Remarkable Axial Substituent Effect on Performance
- PMID: 34963896
- PMCID: PMC8704040
- DOI: 10.1021/acscentsci.1c01066
Modular Design of High-Brightness pH-Activatable Near-Infrared BODIPY Probes for Noninvasive Fluorescence Detection of Deep-Seated Early Breast Cancer Bone Metastasis: Remarkable Axial Substituent Effect on Performance
Abstract
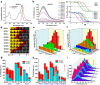
We herein report a series of high-brightness pH-activatable near-infrared (NIR) BODIPY probes for high-contrast intravital imaging of deep-seated early breast cancer bone metastasis by harnessing the axial substituent effect. These probes exhibit tunable pK a, higher brightness, and antiquenching capabilities in aqueous solution, which can be simultaneously adjusted by axial steric substituents. The optimized probe BODO-3 bearing axial dimethyl substituents exhibited a higher pK a value of 5.6 and a brighter NIR fluorescence under tumor acidic pH, showing 10.3-fold and 6.5-fold enhanced brightness (εΦ) at pH 5.5 and 6.5, respectively. Due to the higher brightness, BODO-3 with a brilliant NIR emission at 700 nm allows for deep optical penetrations of 5 and 8 mm at pH 6.5 and 4.5, respectively. Meanwhile, covalent functionalization with glucose (BODO-3-Glu) could further enhance breast cancer and its soft tissue metastasis imaging in vivo. Notably, covalent functionalization with bisphosphonate (BODO-3-PO 3 H 2 ) allowed the successful targeting and visualization of deep-seated bone metastases of breast cancer with a high tumor to normal contrast of 8/1, outperforming X-rays in early detection. This strategy may provide insights for designing high-brightness activatable NIR probes for detecting deep-seated tumors and metastases.
© 2021 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures





References
LinkOut - more resources
Full Text Sources
Miscellaneous

