Biophysical Investigation of the Interplay between the Conformational Species of Domain-Swapped GB1 Amyloid Mutant through Real-Time Monitoring of Amyloid Fibrillation
- PMID: 34963921
- PMCID: PMC8697013
- DOI: 10.1021/acsomega.1c04223
Biophysical Investigation of the Interplay between the Conformational Species of Domain-Swapped GB1 Amyloid Mutant through Real-Time Monitoring of Amyloid Fibrillation
Abstract
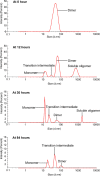
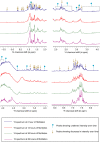
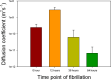
Mutant polypeptide GB1HS#124F26A, which is known to aggregate into amyloid-like fibrils, has been utilized as a model in this study for gaining insights into the mechanism of domain-swapped aggregation through real-time monitoring. Size exclusion with UV monitoring at 280 nm and dynamic light scattering (DLS) profiles through different time points of fibrillation reveal that the dimer transitions into monomeric intermediates during the aggregation, which could further facilitate domain swapping to form amyloid fibrils. The 1D 1H and 2D 1H-13C HSQC nuclear magnetic resonance (NMR) spectra profiling through different time points of fibrillation reveal that there may be some other species present along with the dimer during aggregation which contribute to different trends for the intensity of protons in the spectral peaks. Diffusion NMR reveals changes in the mobility of the dimeric species during the process of aggregation, indicating that the dimer gives rise to other lower molecular weight species midway during aggregation, which further add up to form the oligomers and amyloid fibrils successively. The present work is a preliminary study which explores the possibility of utilizing biophysical methods to gain atomistic level insights into the different stages of aggregation.
© 2021 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

Similar articles
-
Insights into the mechanism of cystatin C oligomer and amyloid formation and its interaction with β-amyloid.J Biol Chem. 2017 Jul 7;292(27):11485-11498. doi: 10.1074/jbc.M117.786558. Epub 2017 May 9. J Biol Chem. 2017. PMID: 28487367 Free PMC article.
-
Runaway domain swapping in amyloid-like fibrils of T7 endonuclease I.Proc Natl Acad Sci U S A. 2006 May 23;103(21):8042-7. doi: 10.1073/pnas.0602607103. Epub 2006 May 12. Proc Natl Acad Sci U S A. 2006. PMID: 16698921 Free PMC article.
-
Mechanism and energy landscape of domain swapping in the B1 domain of protein G.J Mol Biol. 2008 Sep 26;382(1):223-35. doi: 10.1016/j.jmb.2008.06.025. Epub 2008 Jun 17. J Mol Biol. 2008. PMID: 18588900
-
3D domain swapping, protein oligomerization, and amyloid formation.Acta Biochim Pol. 2001;48(4):807-27. Acta Biochim Pol. 2001. PMID: 11995994 Review.
-
Toxic species in amyloid disorders: Oligomers or mature fibrils.Ann Indian Acad Neurol. 2015 Apr-Jun;18(2):138-45. doi: 10.4103/0972-2327.144284. Ann Indian Acad Neurol. 2015. PMID: 26019408 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources

