ADAM17, A Key Player of Cardiac Inflammation and Fibrosis in Heart Failure Development During Chronic Catecholamine Stress
- PMID: 34966735
- PMCID: PMC8710811
- DOI: 10.3389/fcell.2021.732952
ADAM17, A Key Player of Cardiac Inflammation and Fibrosis in Heart Failure Development During Chronic Catecholamine Stress
Abstract
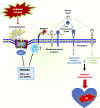
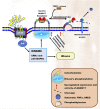
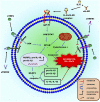
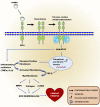
Heart failure development is characterized by persistent inflammation and progressive fibrosis owing to chronic catecholamine stress. In a chronic stress state, elevated catecholamines result in the overstimulation of beta-adrenergic receptors (βARs), specifically β2-AR coupling with Gαi protein. Gαi signaling increases the activation of receptor-stimulated p38 mitogen-activated-protein-kinases (p38 MAPKs) and extracellular signal-regulated kinases (ERKs). Phosphorylation by these kinases is a common way to positively regulate the catalytic activity of A Disintegrin and Metalloprotease 17 (ADAM17), a metalloprotease that has grown much attention in recent years and has emerged as a chief regulatory hub in inflammation, fibrosis, and immunity due to its vital proteolytic activity. ADAM17 cleaves and activates proinflammatory cytokines and fibrotic factors that enhance cardiac dysfunction via inflammation and fibrosis. However, there is limited information on the cardiovascular aspect of ADAM17, especially in heart failure. Hence, this concise review provides a comprehensive insight into the structure of ADAM17, how it is activated and regulated during chronic catecholamine stress in heart failure development. This review highlights the inflammatory and fibrotic roles of ADAM17's substrates; Tumor Necrosis Factor α (TNFα), soluble interleukin-6 receptor (sIL-6R), and amphiregulin (AREG). Finally, how ADAM17-induced chronic inflammation and progressive fibrosis aggravate cardiac dysfunction is discussed.
Keywords: ADAM17; cardiac fibrosis; cardiac inflammation; fibrotic factors; heart failure; metalloenzymes; pro-inflammatory cytokines.
Copyright © 2021 Adu-Amankwaah, Adzika, Adekunle, Ndzie Noah, Mprah, Bushi, Akhter, Huang, Xu, Adzraku, Nadeem and Sun.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
-
- Adekunle A. O., Adzika G. K., Mprah R., Ndzie Noah M. L., Adu-Amankwaah J., Rizvi R., et al. (2021). Predominance of Heart Failure with Preserved Ejection Fraction in Postmenopausal Women: Intra- and Extra-cardiomyocyte Maladaptive Alterations Scaffolded by Estrogen Deficiency. Front. Cell Dev. Biol. 9, 685996. 10.3389/fcell.2021.685996 - DOI - PMC - PubMed
-
- Adu-Amankwaah J., Adzika G. K., Adekunle A. O., Ndzie Noah M. L., Mprah R., Bushi A., et al. (2021a). The Synergy of ADAM17-Induced Myocardial Inflammation and Metabolic Lipids Dysregulation during Acute Stress: New Pathophysiologic Insights into Takotsubo Cardiomyopathy. Front. Cardiovasc. Med. 8, 696413. 10.3389/fcvm.2021.696413 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

