A novel two-factor monosynaptic TRIO tracing method for assessment of circuit integration of hESC-derived dopamine transplants
- PMID: 34971563
- PMCID: PMC8758947
- DOI: 10.1016/j.stemcr.2021.11.014
A novel two-factor monosynaptic TRIO tracing method for assessment of circuit integration of hESC-derived dopamine transplants
Abstract
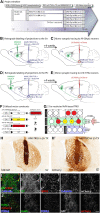
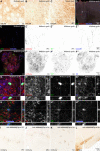
Transplantation in Parkinson's disease using human embryonic stem cell (hESC)-derived dopaminergic (DA) neurons is a promising future treatment option. However, many of the mechanisms that govern their differentiation, maturation, and integration into the host circuitry remain elusive. Here, we engrafted hESCs differentiated toward a ventral midbrain DA phenotype into the midbrain of a preclinical rodent model of Parkinson's disease. We then injected a novel DA-neurotropic retrograde MNM008 adeno-associated virus vector capsid, into specific DA target regions to generate starter cells based on their axonal projections. Using monosynaptic rabies-based tracing, we demonstrated for the first time that grafted hESC-derived DA neurons receive distinctly different afferent inputs depending on their projections. The similarities to the host DA system suggest a previously unknown directed circuit integration. By evaluating the differential host-to-graft connectivity based on projection patterns, this novel approach offers a tool to answer outstanding questions regarding the integration of grafted hESC-derived DA neurons.
Keywords: AAV-MNM008; Cell replacement; Parkinson's disease; animal model; capcid engineering; circuit mapping; dopamine neurons; human embryonic stem cells; monosynaptic tracing; retrograde transport.
Copyright © 2021 The Authors. Published by Elsevier Inc. All rights reserved.
Figures



References
-
- Adler A.F., Cardoso T., Nolbrant S., Mattsson B., Hoban D.B., Jarl U., Wahlestedt J.N., Grealish S., Bjorklund A., Parmar M. hESC-derived dopaminergic transplants integrate into basal Ganglia circuitry in a preclinical model of Parkinson's disease. Cell. Rep. 2019;28:3462–3473.e3465. doi: 10.1016/j.celrep.2019.08.058. - DOI - PMC - PubMed
-
- Aldrin-Kirk P., Heuer A., Wang G., Mattsson B., Lundblad M., Parmar M., Bjorklund T. DREADD modulation of transplanted DA neurons reveals a novel parkinsonian dyskinesia mechanism mediated by the serotonin 5-HT6 receptor. Neuron. 2016;90:955–968. doi: 10.1016/j.neuron.2016.04.017. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

