Molecular mechanisms governing aquaporin relocalisation
- PMID: 34973181
- PMCID: PMC8825993
- DOI: 10.1016/j.bbamem.2021.183853
Molecular mechanisms governing aquaporin relocalisation
Abstract
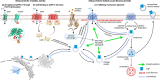
The aquaporins (AQPs) form a family of integral membrane proteins that facilitate the movement of water across biological membrane by osmosis, as well as facilitating the diffusion of small polar solutes. AQPs have been recognised as drug targets for a variety of disorders associated with disrupted water or solute transport, including brain oedema following stroke or trauma, epilepsy, cancer cell migration and tumour angiogenesis, metabolic disorders, and inflammation. Despite this, drug discovery for AQPs has made little progress due to a lack of reproducible high-throughput assays and difficulties with the druggability of AQP proteins. However, recent studies have suggested that targetting the trafficking of AQP proteins to the plasma membrane is a viable alternative drug target to direct inhibition of the water-conducting pore. Here we review the literature on the trafficking of mammalian AQPs with a view to highlighting potential new drug targets for a variety of conditions associated with disrupted water and solute homeostasis.
Keywords: AQP; Aquaporin; Membrane trafficking; Osmosis.
Copyright © 2022 The Authors. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures

Similar articles
-
Recent breakthroughs and future directions in drugging aquaporins.Trends Pharmacol Sci. 2022 Jan;43(1):30-42. doi: 10.1016/j.tips.2021.10.009. Epub 2021 Dec 1. Trends Pharmacol Sci. 2022. PMID: 34863533 Review.
-
Signaling Mechanisms and Pharmacological Modulators Governing Diverse Aquaporin Functions in Human Health and Disease.Int J Mol Sci. 2022 Jan 26;23(3):1388. doi: 10.3390/ijms23031388. Int J Mol Sci. 2022. PMID: 35163313 Free PMC article. Review.
-
Aquaporins: important but elusive drug targets.Nat Rev Drug Discov. 2014 Apr;13(4):259-77. doi: 10.1038/nrd4226. Epub 2014 Mar 14. Nat Rev Drug Discov. 2014. PMID: 24625825 Free PMC article. Review.
-
Molecular Biology of Aquaporins.Adv Exp Med Biol. 2017;969:1-34. doi: 10.1007/978-94-024-1057-0_1. Adv Exp Med Biol. 2017. PMID: 28258563 Review.
-
Insights into plant plasma membrane aquaporin trafficking.Trends Plant Sci. 2013 Jun;18(6):344-52. doi: 10.1016/j.tplants.2012.12.003. Epub 2013 Jan 2. Trends Plant Sci. 2013. PMID: 23291163 Review.
Cited by
-
Capacitation of ram spermatozoa promotes changes in energy metabolism and aquaporin 3 and is affected by individual testosterone variations.Andrology. 2025 May;13(4):921-933. doi: 10.1111/andr.13756. Epub 2024 Sep 6. Andrology. 2025. PMID: 39238428 Free PMC article.
-
Various endurance training intensities improve GFR and Up-regulate AQP2/GSK3β in lithium-induced nephropathic rats.BMC Nephrol. 2025 Feb 6;26(1):60. doi: 10.1186/s12882-025-03997-5. BMC Nephrol. 2025. PMID: 39915719 Free PMC article.
-
Aquaporins alteration revealed kidney damages in cerebral ischemia/reperfusion rats.Heliyon. 2024 May 18;10(10):e31532. doi: 10.1016/j.heliyon.2024.e31532. eCollection 2024 May 30. Heliyon. 2024. PMID: 38807874 Free PMC article.
-
Differential modulation of lung aquaporins among other pathophysiological markers in acute (Cl2 gas) and chronic (carbon nanoparticles, cigarette smoke) respiratory toxicity mouse models.Front Physiol. 2022 Sep 28;13:880815. doi: 10.3389/fphys.2022.880815. eCollection 2022. Front Physiol. 2022. PMID: 36246134 Free PMC article.
-
Critical Role of Aquaporins in Cancer: Focus on Hematological Malignancies.Cancers (Basel). 2022 Aug 29;14(17):4182. doi: 10.3390/cancers14174182. Cancers (Basel). 2022. PMID: 36077720 Free PMC article. Review.
References
-
- Su W., et al. Aquaporins in the kidney: physiology and pathophysiology. Am. J. Physiol. Renal Physiol. 2020;318(1):F193–F203. - PubMed
-
- Kuriyama H., et al. Coordinated regulation of fat-specific and liver-specific glycerol channels, aquaporin adipose and aquaporin 9. Diabetes. 2002;51(10):2915–2921. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- MR/N029453/1/MRC_/Medical Research Council/United Kingdom
- G-1003/PUK_/Parkinson's UK/United Kingdom
- MR/P007058/1/MRC_/Medical Research Council/United Kingdom
- H-1102/PUK_/Parkinson's UK/United Kingdom
- H-1301/PUK_/Parkinson's UK/United Kingdom
- J-0901/PUK_/Parkinson's UK/United Kingdom
- MR/L023784/2/MRC_/Medical Research Council/United Kingdom
- MR/M024962/1/MRC_/Medical Research Council/United Kingdom
- PG/10/54/28460/BHF_/British Heart Foundation/United Kingdom
- K-1003/PUK_/Parkinson's UK/United Kingdom
- G-0801/PUK_/Parkinson's UK/United Kingdom
- BB/D012910/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- BB/P025927/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- MC_EX_MR/N50192X/1/MRC_/Medical Research Council/United Kingdom
LinkOut - more resources
Full Text Sources

