Difficulties, Challenges, and the Learning Curve of Avoiding Complications in Lumbar Endoscopic Spine Surgery
- PMID: 34974418
- PMCID: PMC9421222
- DOI: 10.14444/8161
Difficulties, Challenges, and the Learning Curve of Avoiding Complications in Lumbar Endoscopic Spine Surgery
Abstract
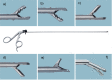
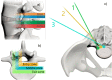
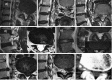
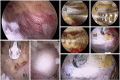
Spinal endoscopy has the stigma of being reserved for only a few surgeons who can figure out how to master the steep learning curve and develop clinical practice settings where endoscopic spine surgery can thrive. In essence, endoscopic treatment of herniated discs specifically and nerve root compression in the lumbar spine in general amounts to replacing traditional open spine surgery protocols with spinal endoscopic surgery techniques. In doing so, the endoscopic spine surgeon must be confident that the degenerative spine's common painful problems can be handled with endoscopic spinal surgery techniques with at least comparable clinical results and complication rates. In this review article, the authors illustrate the difficulties and challenges of the endoscopic lumbar decompression procedure. In addition, they shed light on how to master the learning curve by systematically looking at all sides of the problem, ranging from the ergonomic aspects of the endoscopic platform and its instruments, surgical access planning, challenging clinical scenarios, complications, and sequelae, as well as the training gaps after postgraduate residency and fellowship programs.
Keywords: complications; difficulties; learning curve; lumbar endoscopic surgery; sequalae.
This manuscript is generously published free of charge by ISASS, the International Society for the Advancement of Spine Surgery. Copyright © 2021 ISASS. To see more or order reprints or permissions, see http://ijssurgery.com.
Conflict of interest statement
Declaration of Conflicting Interests: The authors have no direct (employment, stock ownership, grants, patents), or indirect conflicts of interest (honoraria, consultancies to sponsoring organizations, mutual fund ownership, paid expert testimony). The authors are not currently affiliated with or under any consulting agreement with any vendor that the clinical research data conclusion could directly enrich. This manuscript is not meant for or intended to endorse any products or push any agenda other than the associated clinical outcomes with the endoscopic decompression surgery. The motive for compiling this clinically relevant information is by no means created and/or correlated to directly enrich anyone due to its publication. This publication was intended to substantiate contemporary endoscopic spinal surgery concepts to facilitate technology advancements.
Figures





References
-
- Yeung AT, Yeung CA. In-vivo endoscopic visualization of patho-anatomy in painful degenerative conditions of the lumbar spine. Surg Technol Int. 2006;15:243–256. - PubMed
LinkOut - more resources
Full Text Sources
