Outcomes Impacting Quality of Life in Advanced Parkinson's Disease Patients Treated with Levodopa-Carbidopa Intestinal Gel
- PMID: 34974438
- PMCID: PMC9108584
- DOI: 10.3233/JPD-212979
Outcomes Impacting Quality of Life in Advanced Parkinson's Disease Patients Treated with Levodopa-Carbidopa Intestinal Gel
Abstract
Background: It is believed that motor symptoms, including dyskinesia, and non-motor symptoms impact health-related quality of life (HRQoL) in patients with Parkinson's disease (PD), and that improvements in these metrics are correlated.
Objective: Investigate the relationship between HRQoL and measures of PD severity and treatment efficacy, including motor and non-motor symptoms.
Methods: This was a planned investigation of an international, prospective, single-arm, post-marketing observational study of the long-term effectiveness of levodopa-carbidopa intestinal gel (LCIG) in patients with advanced PD. Pearson correlation coefficients (PCC) were calculated for baseline and change from baseline at 12 months between HRQoL and motor and non-motor symptoms.
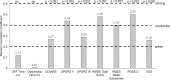
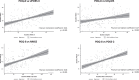
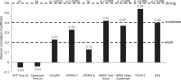
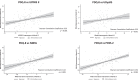
Results: A total of 195 patients were included. At baseline, HRQoL was moderately positively correlated with Activities of Daily Living (UPDRS II, PCC = 0.44), non-motor symptoms (0.48), and measures of sleep (0.50 and 0.40); all p < 0.001. After 12 months of treatment with LCIG, improvements in HRQoL were moderately positively correlated with improvement from baseline in non-motor symptoms (PCC = 0.42), sleep (0.54), and daytime sleepiness (0.40; all p < 0.001), and weakly correlated with improvement in dyskinesia signs and symptoms (PCC = 0.23; p = 0.011). Improvement in HRQoL was not correlated with improvements in OFF time or dyskinesia time.
Conclusion: Both at baseline and for change from baseline at 12 months, HRQoL was correlated with baseline and change from baseline in dyskinesia, Activities of Daily Living, and non-motor symptoms, including sleep; but not with baseline or change in OFF time.
Keywords: Dyskinesia; Parkinson’s disease; health-related quality of life; levodopa-carbidopa intestinal gel; non-motor symptoms.
Conflict of interest statement
Dr Kovács was a study investigator and has received honorarium from AbbVie, Abbott, Boston Scientific, Boehringer Ingelheim, GSK Pharmaceuticals, KRKA, Medtronic, Teva, and UCB Pharma, and has also received honoraria for lecturing at symposia. He has been a consultant for AbbVie, Abbott, KRKA, and Teva. He has received research funding from the Hungarian National Research, Development and Innovation Office; University of Pécs; and Medtronic. Dr Anca-Herschkovitsch is a study investigator and consultant for AbbVie.
Dr Cubo is a study investigator and has received honoraria for consulting from AbbVie, and travel grants from Allergan and Boston Scientific. Dr Davis is a study investigator and has received grant/research support from the Parkinson Foundation and the Peterson Foundation for Parkinsons, and honoraria for consulting from AbbVie. Dr Iansek is a study investigator and has received honorarium for consulting from AbbVie. Dr Siddiqui is a study investigator. He has received research support as a principal investigator from Sunovion Pharma, AbbVie, Boston Scientific Inc, Biogen MA, Theravance Biopharma, Michael J Fox Foundation / Neuropoint Alliance, Impax Laboratories, Neuraly, and Sun Pharma. He has served as a consultant and/or scientific advisor for Boston Scientific and AbbVie. Dr Simu is a study investigator and has received honoraria for lectures at symposia and consultant fees from Teva, AbbVie, Merck, Servier Pharma, AOP Orphan, Boehringer Ingelheim, Sanofi, KRKA, Roche, Novartis, and UCB Pharma. Dr Standaert is a member of the faculty of the University of Alabama at Birmingham and is supported by endowment and University funds. He is an investigator in studies funded by AbbVie; the American Parkinson Disease Association; the Michael J. Fox Foundation for Parkinson Research; Alabama Department of Commerce; the Department of Defense; and NIH grants P50NS108675, R25NS079188, and T32NS095775. He has a clinical practice and is compensated for these activities through the University of Alabama Health Services Foundation. In addition, since 1 January 2020 he has served as a consultant for or received honoraria from AbbVie, Curium, Sutter Health, the Warren Alpert Foundation, the International Parkinson Disease and Movement Disorder Society, Theravance, McGraw Hill, and Sanofi-Aventis.
Dr Chaudhuri is a study investigator and has served as an advisory board member for AbbVie, UCB, GKC, Bial, Cynapsus, Lobsor, Stada, Medtronic, Zambon, Profile, Sunovion, Roche, Therevance, Scion, Britannia, Acadia, and 4D. He received honoraria for lectures from AbbVie, Britannia, UCB, Zambon, Novartis, Boeringer Ingelheim, Bial, Kyowa Kirin, and SK Pharma. He has received grants (investigator initiated) from Britannia Pharmaceuticals, AbbVie, UCB, GKC, and Bial; and academic grants from EU, IMI EU, Horizon 2020, Parkinson’s UK, NIHR, PDNMG, EU (Horizon 2020), Kirby Laing Foundation, NPF, MRC, and Wellcome Trust. He receives royalties from Oxford University Press and holds intellectual property rights for the King’s Parkinson’s Pain Scale and Parkinson’s Disease Sleep Scale. Dr Bourgeois has received honoraria for consulting from AbbVie. Dr Pontieri is a member of the faculty at the Sapienza University of Rome and has a clinical practice at Sant’Andrea Hospital in Rome. He is also Senior Research Neurologist at the Fondazione Santa Lucia, IRCCS, Rome. He is an investigator in studies funded by AbbVie, Zambon, and the Italian Ministry of Health and has served as consultant for or received honoraria from AbbVie, Zambon, FB Health, Bial, and Lundbeck. Dr Aldred has received honoraria from Abbott Laboratories, AbbVie, Accorda, Allergan, Medtronic, Boston Scientific, Teva, and US World Meds. He has received research funding for studies sponsored by AbbVie, Acadia, Annexon, Amneal/Impax, Biogen, Boston Scientific, Centogene, Denali, Massachusetts General Hospital, Neuroderm, Neurocrine, Northwestern University, Sunovion, Triplet Therapeutics, and Theravance. He is a scientific advisor for AbbVie, Allergan, and Boston Scientific. Dr Bergmann, Dr Gao, and Dr Kukreja are employees of AbbVie and may hold AbbVie stock and/or stock options.
Figures

References
-
- Rao SS, Hofmann LA, Shakil A (2006) Parkinson’s disease: Diagnosis and treatment. Am Fam Physician 74, 2046–2054. - PubMed
-
- Miyasaki JM, Martin W, Suchowersky O, Weiner WJ, Lang AE (2002) Practice parameter: Initiation of treatment for Parkinson’s disease: An evidence-based review: Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 58, 11–17. - PubMed
-
- Fox SH, Katzenschlager R, Lim SY, Barton B, de Bie RMA, Seppi K, Coelho M, Sampaio C, Movement Disorder Society Evidence-Based Medicine Committee (2018) International Parkinson and Movement Disorder Society evidence-based medicine review: Update on treatments for the motor symptoms of Parkinson’s disease. Mov Disord 33, 1248–1266. - PubMed
-
- Antonini A, Poewe W, Chaudhuri KR, Jech R, Pickut B, Pirtošek Z, Szasz J, Valldeoriola F, Winkler C, Bergmann L, Yegin A, Onuk K, Barch D, Odin P (2017) Levodopa-carbidopa intestinal gel in advanced Parkinson’s: Final results of the GLORIA registry. Parkinsonism Relat Disord 45, 13–20. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

