B cells do not play a role in vaccine-mediated immunity against Marek's disease
- PMID: 34977551
- PMCID: PMC8686028
- DOI: 10.1016/j.jvacx.2021.100128
B cells do not play a role in vaccine-mediated immunity against Marek's disease
Abstract
Background: Marek's disease virus (MDV), a highly oncogenic α-herpesvirus, is the etiological agent of Marek's disease (MD) in chickens. The antiviral activity of vaccine-induced immunity against MD reduces the level of early cytolytic infection, production of cell-free virions in the feather follicle epithelial cells (FFE), and lymphoma formation. Despite the success of several vaccines that have greatly reduced the economic losses from MD, the mechanism of vaccine-induced immunity is poorly understood.
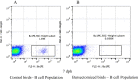
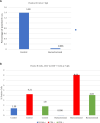
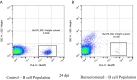
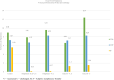
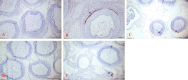
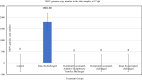
Methods: To provide insight into possible role of B cells in vaccine-mediated protection, we bursectomized birds on day of hatch and vaccinated them eight days later. The birds were challenged 10 days post vaccination with or without receiving adoptive lymphocytes from age-matched control birds prior to inoculation. The study also included vaccinated/challenged and non-vaccinated challenged intact birds. Flowcytometric analysis of PBMN cells were conducted twice post bursectomy to confirm B cell depletion and assess the effect of surgery on T cell population. Immunohistochemical analysis and viral genome copy number assessment in the skin samples at termination was performed to measure the replication rate of MDV in the FFE of the skin tissues of the challenged birds.
Results: The non-vaccinated/challenged birds developed typical clinical signs of MD while the vaccinated/challenged and bursectomized, vaccinated/challenged groups with or without adoptive lymphocyte transfer, were fully protected with no sign of transient paralysis, weight loss, or T cell lymphomas. Immunohistochemical analysis and viral genome copy number evaluation in the skin samples revealed that unlike the vaccinated/challenged birds a significant number of virus particles were produced in the FFE of the non-vaccinated/challenged birds at termination. In the bursectomized, vaccinated/challenged groups, only a few replicating virions were detected in the skin of birds that received adoptive lymphocytes prior to challenge.
Conclusions: The study shows that B cells do not play a critical role in MD vaccine-mediated immunity.
Keywords: B cells; Bursectomy; Marek’s disease; Marek’s disease virus; Vaccination.
© 2021 The Authors. Published by Elsevier Ltd.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures
Similar articles
-
Role of T Cells in Vaccine-Mediated Immunity against Marek's Disease.Viruses. 2023 Feb 28;15(3):648. doi: 10.3390/v15030648. Viruses. 2023. PMID: 36992357 Free PMC article.
-
Towards a mechanistic understanding of the synergistic response induced by bivalent Marek's disease vaccines to prevent lymphomas.Vaccine. 2019 Oct 8;37(43):6397-6404. doi: 10.1016/j.vaccine.2019.09.003. Epub 2019 Sep 9. Vaccine. 2019. PMID: 31515142
-
RNA-seq analysis of viral gene expression in the skin of Marek's disease virus infected chickens.Vet Immunol Immunopathol. 2019 Jul;213:109882. doi: 10.1016/j.vetimm.2019.109882. Epub 2019 Jun 18. Vet Immunol Immunopathol. 2019. PMID: 31307672
-
Prevention of Marek's disease: a review.Cancer Res. 1976 Feb;36(2 pt 2):696-700. Cancer Res. 1976. PMID: 766959 Review.
-
Pathogenesis of Marek's disease (MD) and possible mechanisms of immunity induced by MD vaccine.J Vet Med Sci. 1998 Jan;60(1):1-8. doi: 10.1292/jvms.60.1. J Vet Med Sci. 1998. PMID: 9492353 Review.
Cited by
-
Immune escape of avian oncogenic Marek's disease herpesvirus and antagonistic host immune responses.NPJ Vaccines. 2024 Jun 15;9(1):109. doi: 10.1038/s41541-024-00905-0. NPJ Vaccines. 2024. PMID: 38879650 Free PMC article. Review.
-
Development of an ELISPOT assay for numerating IFN-γ-secreting T cells in chicken using novel monoclonal antibodies.Poult Sci. 2025 Jul 10;104(10):105524. doi: 10.1016/j.psj.2025.105524. Online ahead of print. Poult Sci. 2025. PMID: 40669239 Free PMC article.
-
Marek's disease virus-specific T cells proliferate, express antiviral cytokines but have impaired degranulation response.Front Immunol. 2022 Sep 15;13:973762. doi: 10.3389/fimmu.2022.973762. eCollection 2022. Front Immunol. 2022. PMID: 36189228 Free PMC article.
-
Role of T Cells in Vaccine-Mediated Immunity against Marek's Disease.Viruses. 2023 Feb 28;15(3):648. doi: 10.3390/v15030648. Viruses. 2023. PMID: 36992357 Free PMC article.
-
First report of co-infections of Marek's disease virus and chicken infectious anaemia virus in poultry flocks in Nigeria.Vet Anim Sci. 2024 Feb 13;23:100339. doi: 10.1016/j.vas.2024.100339. eCollection 2024 Mar. Vet Anim Sci. 2024. PMID: 38406258 Free PMC article.
References
-
- Calnek B.W. Pathogenesis of Marek's disease virus infection. Curr Top Microbiol Immunol. 2001;255:25–55. - PubMed
-
- Barrow A.D., Burgess S.C., Baigent S.J., Howes K., Nair V.K. Infection of macrophages by a lymphotropic herpesvirus: a new tropism for Marek's disease virus. J Gen Virol. 2003;84:2635–2645. - PubMed
-
- Osterrieder N., Kamil J.P., Schumacher D., Tischer B.K., Trapp S. Marek's disease virus: from miasma to model. Nat Rev Microbiol. 2006;4(4):283–294. - PubMed
LinkOut - more resources
Full Text Sources

