B cell class switching in intestinal immunity in health and disease
- PMID: 34978077
- PMCID: PMC9285483
- DOI: 10.1111/sji.13139
B cell class switching in intestinal immunity in health and disease
Abstract
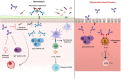
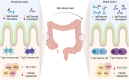
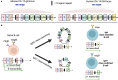
The gastrointestinal tract is colonized by trillions of commensal microorganisms that collectively form the microbiome and make essential contributions to organism homeostasis. The intestinal immune system must tolerate these beneficial commensals, whilst preventing pathogenic organisms from systemic spread. Humoral immunity plays a key role in this process, with large quantities of immunoglobulin (Ig)A secreted into the lumen on a daily basis, regulating the microbiome and preventing bacteria from encroaching on the epithelium. However, there is an increasing appreciation of the role of IgG antibodies in intestinal immunity, including beneficial effects in neonatal immune development, pathogen and tumour resistance, but also of pathological effects in driving chronic inflammation in inflammatory bowel disease (IBD). These antibody isotypes differ in effector function, with IgG exhibiting more proinflammatory capabilities compared with IgA. Therefore, the process that leads to the generation of different antibody isotypes, class-switch recombination (CSR), requires careful regulation and is orchestrated by the immunological cues generated by the prevalent local challenge. In general, an initiating signal such as CD40 ligation on B cells leads to the induction of activation-induced cytidine deaminase (AID), but a second cytokine-mediated signal determines which Ig heavy chain is expressed. Whilst the cytokines driving intestinal IgA responses are well-studied, there is less clarity on how IgG responses are generated in the intestine, and how these cues might become dysfunctional in IBD. Here, we review the key mechanisms regulating class switching to IgA vs IgG in the intestine, processes that could be therapeutically manipulated in infection and IBD.
Keywords: B cells; antibodies; intestinal immunity.
© 2022 The Authors. Scandinavian Journal of Immunology published by John Wiley & Sons Ltd on behalf of The Scandinavian Foundation for Immunology.
Conflict of interest statement
The authors declare no competing interests.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

