Acute normobaric hypoxia blunts contraction-mediated mTORC1- and JNK-signaling in human skeletal muscle
- PMID: 34984845
- PMCID: PMC9285439
- DOI: 10.1111/apha.13771
Acute normobaric hypoxia blunts contraction-mediated mTORC1- and JNK-signaling in human skeletal muscle
Abstract
Aim: Hypoxia has been shown to reduce resistance exercise-induced stimulation of protein synthesis and long-term gains in muscle mass. However, the mechanism whereby hypoxia exerts its effect is not clear. Here, we examine the effect of acute hypoxia on the activity of several signalling pathways involved in the regulation of muscle growth following a bout of resistance exercise.
Methods: Eight men performed two sessions of leg resistance exercise in normoxia or hypoxia (12% O2 ) in a randomized crossover fashion. Muscle biopsies were obtained at rest and 0, 90,180 minutes after exercise. Muscle analyses included levels of signalling proteins and metabolites associated with energy turnover.
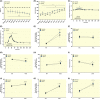
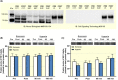
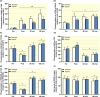
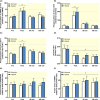
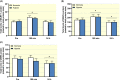
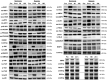
Results: Exercise during normoxia induced a 5-10-fold increase of S6K1Thr389 phosphorylation throughout the recovery period, but hypoxia blunted the increases by ~50%. Phosphorylation of JNKThr183/Tyr185 and the JNK target SMAD2Ser245/250/255 was increased by 30- to 40-fold immediately after the exercise in normoxia, but hypoxia blocked almost 70% of the activation. Throughout recovery, phosphorylation of JNK and SMAD2 remained elevated following the exercise in normoxia, but the effect of hypoxia was lost at 90-180 minutes post-exercise. Hypoxia had no effect on exercise-induced Hippo or autophagy signalling and ubiquitin-proteasome related protein levels. Nor did hypoxia alter the changes induced by exercise in high-energy phosphates, glucose 6-P, lactate or phosphorylation of AMPK or ACC.
Conclusion: We conclude that acute severe hypoxia inhibits resistance exercise-induced mTORC1- and JNK signalling in human skeletal muscle, effects that do not appear to be mediated by changes in the degree of metabolic stress in the muscle.
Keywords: FSR; Hippo pathway; deuterium oxide; muscle metabolites; oxygen.
© 2022 The Authors. Acta Physiologica published by John Wiley & Sons Ltd on behalf of Scandinavian Physiological Society.
Conflict of interest statement
The authors declare that they have no conflict of interests.
Figures



Comment in
-
The Art of Planning-How many samples are enough?Acta Physiol (Oxf). 2022 Feb;234(2):e13746. doi: 10.1111/apha.13746. Epub 2021 Dec 20. Acta Physiol (Oxf). 2022. PMID: 34907659 No abstract available.
-
How hypoxia blunts protein synthesis activation after resistance exercise.Acta Physiol (Oxf). 2022 Feb;234(2):e13747. doi: 10.1111/apha.13747. Epub 2021 Dec 29. Acta Physiol (Oxf). 2022. PMID: 34931453 No abstract available.
References
-
- McKendry J, Breen L, Shad BJ, Greig CA. Muscle morphology and performance in master athletes: a systematic review and meta‐analyses. Ageing Res Rev. 2018;45:62‐82. - PubMed
-
- Filippin LI, Teixeira VN, da Silva MP, Miraglia F, da Silva FS. Sarcopenia: a predictor of mortality and the need for early diagnosis and intervention. Aging Clin Exp Res. 2015;27(3):249‐254. - PubMed
-
- Hoppeler H, Kleinert E, Schlegel C, et al. Morphological adaptations of human skeletal muscle to chronic hypoxia. Int J Sports Med. 1990;11(Suppl 1):S3‐S9. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

