Antiviral CD19+CD27+ Memory B Cells Are Associated with Protection from Recurrent Asymptomatic Ocular Herpesvirus Infection
- PMID: 34985998
- PMCID: PMC8906405
- DOI: 10.1128/jvi.02057-21
Antiviral CD19+CD27+ Memory B Cells Are Associated with Protection from Recurrent Asymptomatic Ocular Herpesvirus Infection
Abstract
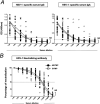
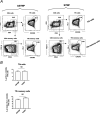
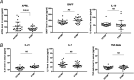
Reactivation of herpes simplex virus 1 (HSV-1) from latently infected neurons of the trigeminal ganglia (TG) leads to blinding recurrent herpetic disease in symptomatic (SYMP) individuals. Although the role of T cells in herpes immunity seen in asymptomatic (ASYMP) individuals is heavily explored, the role of B cells is less investigated. In the present study, we evaluated whether B cells are associated with protective immunity against recurrent ocular herpes. The frequencies of circulating HSV-specific memory B cells and of memory follicular helper T cells (CD4+ Tfh cells), which help B cells produce antibodies, were compared between HSV-1-infected SYMP and ASYMP individuals. The levels of IgG/IgA and neutralizing antibodies were compared in SYMP and ASYMP individuals. We found that (i) the ASYMP individuals had increased frequencies of HSV-specific CD19+CD27+ memory B cells, and (ii) high frequencies of HSV-specific switched IgG+CD19+CD27+ memory B cells detected in ASYMP individuals were directly proportional to high frequencies of CD45R0+CXCR5+CD4+ memory Tfh cells. However, no differences were detected in the level of HSV-specific IgG/IgA antibodies in SYMP and ASYMP individuals. Using the UV-B-induced HSV-1 reactivation mouse model, we found increased frequencies of HSV-specific antibody-secreting plasma HSV-1 gD+CD138+ B cells within the TG and circulation of ASYMP mice compared to those of SYMP mice. In contrast, no significant differences in the frequencies of B cells were found in the cornea, spleen, and bone-marrow. Our findings suggest that circulating antibody-producing HSV-specific memory B cells recruited locally to the TG may contribute to protection from symptomatic recurrent ocular herpes. IMPORTANCE Reactivation of herpes simplex virus 1 (HSV-1) from latently infected neurons of the trigeminal ganglia (TG) leads to blinding recurrent herpetic disease in symptomatic (SYMP) individuals. Although the role of T cells in herpes immunity against blinding recurrent herpetic disease is heavily explored, the role of B cells is less investigated. In the present study, we found that in both asymptomatic (ASYMP) individuals and ASYMP mice, there were increased frequencies of HSV-specific memory B cells that were directly proportional to high frequencies of memory Tfh cells. Moreover, following UV-B-induced reactivation, we found increased frequencies of HSV-specific antibody-secreting plasma B cells within the TG and circulation of ASYMP mice compared to those of SYMP mice. Our findings suggest that circulating antibody-producing HSV-specific memory B cells recruited locally to the TG may contribute to protection from recurrent ocular herpes.
Keywords: B memory cells; asymptomatic herpes; ocular herpes; plasma cells; virus-specific B cell.
Conflict of interest statement
The authors declare no conflict of interest.
The authors have declared that no conflict of interest exists.
Figures




Similar articles
-
Human Asymptomatic Epitopes Identified from the Herpes Simplex Virus Tegument Protein VP13/14 (UL47) Preferentially Recall Polyfunctional Effector Memory CD44high CD62Llow CD8+ TEM Cells and Protect Humanized HLA-A*02:01 Transgenic Mice against Ocular Herpesvirus Infection.J Virol. 2017 Jan 3;91(2):e01793-16. doi: 10.1128/JVI.01793-16. Print 2017 Jan 15. J Virol. 2017. PMID: 27847359 Free PMC article.
-
Phenotypic and functional characterization of herpes simplex virus glycoprotein B epitope-specific effector and memory CD8+ T cells from symptomatic and asymptomatic individuals with ocular herpes.J Virol. 2015 Apr;89(7):3776-92. doi: 10.1128/JVI.03419-14. Epub 2015 Jan 21. J Virol. 2015. PMID: 25609800 Free PMC article.
-
Unique Type I Interferon, Expansion/Survival Cytokines, and JAK/STAT Gene Signatures of Multifunctional Herpes Simplex Virus-Specific Effector Memory CD8+ TEM Cells Are Associated with Asymptomatic Herpes in Humans.J Virol. 2019 Feb 5;93(4):e01882-18. doi: 10.1128/JVI.01882-18. Print 2019 Feb 15. J Virol. 2019. PMID: 30487281 Free PMC article.
-
Recurrent herpetic stromal keratitis in mice: a model for studying human HSK.Clin Dev Immunol. 2012;2012:728480. doi: 10.1155/2012/728480. Epub 2012 Apr 23. Clin Dev Immunol. 2012. PMID: 22593769 Free PMC article. Review.
-
[Battle with herpes for 37 years].Nippon Ganka Gakkai Zasshi. 2015 Mar;119(3):145-66; discussion 167. Nippon Ganka Gakkai Zasshi. 2015. PMID: 25854108 Review. Japanese.
Cited by
-
HSV-1 0∆NLS vaccine elicits a robust B lymphocyte response and preserves vision without HSV-1 glycoprotein M or thymidine kinase recognition.Sci Rep. 2022 Sep 23;12(1):15920. doi: 10.1038/s41598-022-20180-0. Sci Rep. 2022. PMID: 36151255 Free PMC article.
-
Cytokines and chemokines: The vital role they play in herpes simplex virus mucosal immunology.Front Immunol. 2022 Sep 23;13:936235. doi: 10.3389/fimmu.2022.936235. eCollection 2022. Front Immunol. 2022. PMID: 36211447 Free PMC article. Review.
-
Herpes simplex keratitis: A brief clinical overview.World J Virol. 2024 Mar 25;13(1):89934. doi: 10.5501/wjv.v13.i1.89934. World J Virol. 2024. PMID: 38616855 Free PMC article. Review.
-
Single-cell RNA sequencing reveals the diversity of the immunological landscape response to genital herpes.Virol Sin. 2024 Dec;39(6):860-874. doi: 10.1016/j.virs.2024.10.003. Epub 2024 Oct 18. Virol Sin. 2024. PMID: 39426602 Free PMC article.
-
Immunological Control of Herpes Simplex Virus Type 1 Infection: A Non-Thermal Plasma-Based Approach.Viruses. 2025 Apr 23;17(5):600. doi: 10.3390/v17050600. Viruses. 2025. PMID: 40431612 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

