STING orchestrates the crosstalk between polyunsaturated fatty acid metabolism and inflammatory responses
- PMID: 34986331
- PMCID: PMC8733004
- DOI: 10.1016/j.cmet.2021.12.007
STING orchestrates the crosstalk between polyunsaturated fatty acid metabolism and inflammatory responses
Abstract
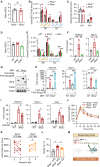
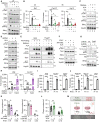
Concerted alteration of immune and metabolic homeostasis underlies several inflammation-related pathologies, ranging from metabolic syndrome to infectious diseases. Here, we explored the coordination of nucleic acid-dependent inflammatory responses and metabolic homeostasis. We reveal that the STING (stimulator of interferon genes) protein regulates metabolic homeostasis through inhibition of the fatty acid desaturase 2 (FADS2) rate-limiting enzyme in polyunsaturated fatty acid (PUFA) desaturation. STING ablation and agonist-mediated degradation increased FADS2-associated desaturase activity and led to accumulation of PUFA derivatives that drive thermogenesis. STING agonists directly activated FADS2-dependent desaturation, promoting metabolic alterations. PUFAs in turn inhibited STING, thereby regulating antiviral responses and contributing to resolving STING-associated inflammation. Thus, we have unveiled a negative regulatory feedback loop between STING and FADS2 that fine-tunes inflammatory responses. Our results highlight the role of metabolic alterations in human pathologies associated with aberrant STING activation and STING-targeting therapies.
Keywords: FADS2; STING; cGAS; cytosolic DNA; delta-6 Desaturase; inflammation; interferon responses; metabolism; nucleic acid immunity; polyunsaturated fatty acids.
Copyright © 2021 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests J.M. is a joint PhD student in Azelead, a startup company, and the Laguette laboratory. All other authors declare no competing interests.
Figures





Comment in
-
Multiplexed targeted analysis of polyunsaturated fatty acids and oxylipins using liquid chromatography-tandem mass spectrometry.STAR Protoc. 2023 Sep 15;4(3):102226. doi: 10.1016/j.xpro.2023.102226. Epub 2023 Aug 17. STAR Protoc. 2023. PMID: 37597187 Free PMC article.
References
-
- Ablasser A., Hemmerling I., Schmid-Burgk J.L., Behrendt R., Roers A., Hornung V. TREX1 deficiency triggers cell-autonomous immunity in a cGAS-dependent manner. J. Immunol. 2014;192:5993–5997. - PubMed
-
- Belchior T., Paschoal V.A., Magdalon J., Chimin P., Farias T.M., Chaves-Filho A.B., Gorjão R., St-Pierre P., Miyamoto S., Kang J.X., et al. Omega-3 fatty acids protect from diet-induced obesity, glucose intolerance, and adipose tissue inflammation through PPARγ-dependent and PPARγ-independent actions. Mol. Nutr. Food Res. 2015;59:957–967. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

