RNA profiles reveal signatures of future health and disease in pregnancy
- PMID: 34987224
- PMCID: PMC8770117
- DOI: 10.1038/s41586-021-04249-w
RNA profiles reveal signatures of future health and disease in pregnancy
Abstract
Maternal morbidity and mortality continue to rise, and pre-eclampsia is a major driver of this burden1. Yet the ability to assess underlying pathophysiology before clinical presentation to enable identification of pregnancies at risk remains elusive. Here we demonstrate the ability of plasma cell-free RNA (cfRNA) to reveal patterns of normal pregnancy progression and determine the risk of developing pre-eclampsia months before clinical presentation. Our results centre on comprehensive transcriptome data from eight independent prospectively collected cohorts comprising 1,840 racially diverse pregnancies and retrospective analysis of 2,539 banked plasma samples. The pre-eclampsia data include 524 samples (72 cases and 452 non-cases) from two diverse independent cohorts collected 14.5 weeks (s.d., 4.5 weeks) before delivery. We show that cfRNA signatures from a single blood draw can track pregnancy progression at the placental, maternal and fetal levels and can robustly predict pre-eclampsia, with a sensitivity of 75% and a positive predictive value of 32.3% (s.d., 3%), which is superior to the state-of-the-art method2. cfRNA signatures of normal pregnancy progression and pre-eclampsia are independent of clinical factors, such as maternal age, body mass index and race, which cumulatively account for less than 1% of model variance. Further, the cfRNA signature for pre-eclampsia contains gene features linked to biological processes implicated in the underlying pathophysiology of pre-eclampsia.
© 2022. The Author(s).
Conflict of interest statement
M. Rasmussen, M. Reddy, R.N., J.C.-S., A.K., T.B., F.S., M.T., E.P.S.G., J.L., M.L., E.N., M.J., M.A.E., M.D., S.R.Q. and T.M. have an equity interest in Mirvie. All cohort contributors were compensated for sample collection and/or shipping. T.M. serves on the scientific advisory board for Mirvie, NxPrenatal, Momenta Pharmaceuticals and Hoffmann–La Roche. M. Rasmussen, M. Reddy, R.N., J.C.-S., A.K., T.B., F.S., M.T., E.P.S.G., J.L., M.L., E.N., M.J., M.A.E., S.R.Q., M.K.S. and D.A.S. are inventors on patent applications (US20170145509A1, US9937182B2 and EP2954324A1) that cover the detection, diagnosis or treatment of pregnancy complications.
Figures



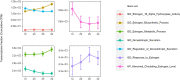
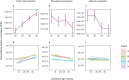


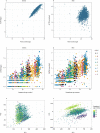
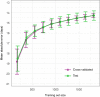

Comment in
-
A blood test to predict complications of pregnancy.Nature. 2022 Jan;601(7893):319-320. doi: 10.1038/d41586-021-03801-y. Nature. 2022. PMID: 34987208 No abstract available.
-
Detecting pregnancy complications from blood.Nat Rev Genet. 2022 Mar;23(3):136. doi: 10.1038/s41576-022-00453-0. Nat Rev Genet. 2022. PMID: 35082441 No abstract available.
-
Cell-free plasma RNA signatures as a surrogate biomarker of pregnancy health.Med. 2022 Feb 11;3(2):90-92. doi: 10.1016/j.medj.2022.01.010. Med. 2022. PMID: 35590212
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

