Assessment of first-line eradication treatment in Greece: data from the European Registry on Helicobacter pylori management (Hp-EuReg)
- PMID: 34987287
- PMCID: PMC8713343
- DOI: 10.20524/aog.2021.0670
Assessment of first-line eradication treatment in Greece: data from the European Registry on Helicobacter pylori management (Hp-EuReg)
Abstract
Background: Helicobacter pylori (H. pylori) is the most common chronic bacterial infection. Its management has to rely on local effectiveness, given the geographical variability of bacterial antibiotic resistance. We evaluated treatment effectiveness in naïve patients in Greece, as part of the European Registry on the management of H. pylori (Hp-EuReg).
Methods: Patients were registered in the AEG-REDCap Electronic Case Report Form from 2013-2020. All cases with a first-line treatment were included. Modified intention-to-treat (mITT) analysis was used.
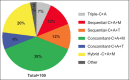
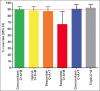
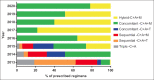
Results: A total of 547 patients from 5 medical institutions were treated with the following regimens: concomitant with proton pump inhibitors (PPIs), clarithromycin, amoxicillin and metronidazole (concomitant-C+A+M) (38%); hybrid with PPI, clarithromycin, amoxicillin and metronidazole (hybrid-C+A+M) (20%); sequential with PPI, clarithromycin, amoxicillin and tinidazole (sequential-C+A+T) (12%); sequential with PPI, clarithromycin, amoxicillin and metronidazole (sequential-C+A+M) (12%); concomitant with PPI, clarithromycin, amoxicillin and tinidazole (concomitant-C+A+T) (8%); triple with PPI, clarithromycin and amoxicillin (triple-C+A) (7%); and other (3%). Overall compliance was 99%. Triple-C+A, sequential-C+A+T, sequential-C+A+M and concomitant-C+A+T were used from 2013-2015. The respective mITT cure rates (95% confidence interval) were 92% (78-98), 87% (76-94), 67% (54-78) and 91% (79-98). Since 2015, patients were also treated with concomitant-C+A+M and hybrid-C+A+M regimens, with respective mITT cure rates of 90% (85-94) and 88% (80.5-94). Adverse events were reported by 31% of the patients, dysgeusia being the most frequent (15%).
Conclusions: "Optimized" H. pylori therapies should achieve cure rates over 90%. In Greece, at present, only non-bismuth quadruple concomitant regimens achieve this target and can be recommended as first-line treatment.
Keywords: Greece; Helicobacter pylori; Hp-EuReg; treatment.
Copyright: © Hellenic Society of Gastroenterology.
Conflict of interest statement
Conflicts of interest: Dr. Megraud received research support from Aptalis Pharma, bioMerieux and Mobidiag and is a consultant for Phathom Pharmaceuticals. Dr. Gisbert has served as speaker, consultant, and advisory member for, or has received research funding from Mayoly, Allergan, Diasorin, Phathom and Gebro Pharma. Dr Nyssen has received research funding from Mayoly and Allergan
Figures
Similar articles
-
First-Line Prescriptions and Effectiveness of Helicobacter pylori Eradication Treatment in Ireland over a 10-Year Period: Data from the European Registry on Helicobacter pylori Management (Hp-EuReg).Antibiotics (Basel). 2025 Jul 5;14(7):680. doi: 10.3390/antibiotics14070680. Antibiotics (Basel). 2025. PMID: 40723983 Free PMC article.
-
[Effectiveness of empirical Helicobacter pylori eradication therapy with furazolidone in Russia: results from the European Registry on Helicobacter pylori Management (Hp-EuReg)].Ter Arkh. 2023 Mar 30;95(2):120-129. doi: 10.26442/00403660.2023.02.202107. Ter Arkh. 2023. PMID: 37167127 Russian.
-
Second-line rescue triple therapy with levofloxacin after failure of non-bismuth quadruple "sequential" or "concomitant" treatment to eradicate H. pylori infection.Scand J Gastroenterol. 2013 Jun;48(6):652-6. doi: 10.3109/00365521.2013.786132. Epub 2013 Apr 5. Scand J Gastroenterol. 2013. PMID: 23556551 Clinical Trial.
-
Review article: non-bismuth quadruple (concomitant) therapy for eradication of Helicobater pylori.Aliment Pharmacol Ther. 2011 Sep;34(6):604-17. doi: 10.1111/j.1365-2036.2011.04770.x. Epub 2011 Jul 11. Aliment Pharmacol Ther. 2011. PMID: 21745241 Review.
-
A new look at anti-Helicobacter pylori therapy.World J Gastroenterol. 2011 Sep 21;17(35):3971-5. doi: 10.3748/wjg.v17.i35.3971. World J Gastroenterol. 2011. PMID: 22046084 Free PMC article. Review.
Cited by
-
Equivalence Trial of the Non-Bismuth 10-Day Concomitant and 14-Day Hybrid Therapies for Helicobacter pylori Eradication in High Clarithromycin Resistance Areas.Antibiotics (Basel). 2024 Mar 20;13(3):280. doi: 10.3390/antibiotics13030280. Antibiotics (Basel). 2024. PMID: 38534715 Free PMC article.
References
-
- Liou JM, Malfertheiner P, Lee YC, et al. Asian Pacific Alliance on Helicobacter and Microbiota (APAHAM) Screening and eradication of Helicobacter pylori for gastric cancer prevention:the Taipei global consensus. Gut. 2020;69:2093–2112. - PubMed
-
- McNicholl AG, O'Morain CA, Megraud F, Gisbert JP As Scientific Committee of the Hp-Eureg on Behalf of the National Coordinators. Protocol of the European Registry on the management of Helicobacter pylori infection (Hp-EuReg) Helicobacter. 2019;24:e12630. - PubMed
-
- Nyssen OP, Bordin D, Tepes B, et al. Hp-EuReg Investigators. European Registry on Helicobacter pylori management (Hp-EuReg):patterns and trends in first-line empirical eradication prescription and outcomes of 5 years and 21,533 patients. Gut. 2021;70:40–54. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
