Extracellular Vesicles: Potential Role in Remote Signaling and Inflammation in Trypanosoma cruzi-Triggered Disease
- PMID: 34988085
- PMCID: PMC8721122
- DOI: 10.3389/fcell.2021.798054
Extracellular Vesicles: Potential Role in Remote Signaling and Inflammation in Trypanosoma cruzi-Triggered Disease
Abstract
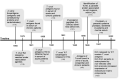
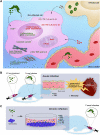
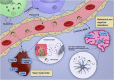
Extracellular vesicles (EVs) act as cell communicators and immune response modulators and may be employed as disease biomarkers and drug delivery systems. In infectious diseases, EVs can be released by the pathogen itself or by the host cells (infected or uninfected), potentially impacting the outcome of the immune response and pathological processes. Chagas disease (CD) is caused by infection by the protozoan Trypanosoma cruzi and is the main cause of heart failure in endemic areas. This illness attracted worldwide attention due to the presence of symptomatic seropositive subjects in North America, Asia, Oceania, and Europe. In the acute phase of infection, nonspecific signs, and symptoms contribute to miss diagnosis and early etiological treatment. In this phase, the immune response is crucial for parasite control; however, parasite persistence, dysregulated immune response, and intrinsic tissue factors may contribute to the pathogenesis of chronic CD. Most seropositive subjects remain in the indeterminate chronic form, and from 30 to 40% of the subjects develop cardiac, digestive, or cardio-digestive manifestations. Identification of EVs containing T. cruzi antigens suggests that these vesicles may target host cells and regulate cellular processes and the immune response by molecular mechanisms that remain to be determined. Parasite-released EVs modulate the host-parasite interplay, stimulate intracellular parasite differentiation and survival, and promote a regulatory cytokine profile in experimental models of CD. EVs derived from the parasite-cell interaction inhibit complement-mediated parasite lysis, allowing evasion. EVs released by T. cruzi-infected cells also regulate surrounding cells, maintaining a proinflammatory profile. After a brief review of the basic features of EVs, the present study focuses on potential participation of T. cruzi-secreted EVs in cell infection and persistence of low-grade parasite load in the chronic phase of infection. We also discuss the role of EVs in shaping the host immune response and in pathogenesis and progression of CD.
Keywords: Chagas disease; Trypanosoma cruzi; extracellular vesicles; immune response; inflammation.
Copyright © 2021 Dantas-Pereira, Menna-Barreto and Lannes-Vieira.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Abuin G., Colli W., Alves M. J. (1996). Turnover and Shedding of the Tc-85 Surface Glycoprotein of Trypanosoma Cruzi Trypomastigotes. Braz. J. Med. Biol. Res. 29 (3), 335–341. - PubMed
-
- Aliberti J. C., Cardoso M. A., Martins G. A., Gazzinelli R. T., Vieira L. Q., Silva J. S. (1996). Interleukin-12 Mediates Resistance to Trypanosoma Cruzi in Mice and Is Produced by Murine Macrophages in Response to Live Trypomastigotes. Infect. Immun. 64 (6), 1961–1967. 10.1128/iai.64.6.1961-1967.1996 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

