Association of Fecal and Plasma Levels of Short-Chain Fatty Acids With Gut Microbiota and Clinical Severity in Patients With Parkinson Disease
- PMID: 34996879
- PMCID: PMC8883514
- DOI: 10.1212/WNL.0000000000013225
Association of Fecal and Plasma Levels of Short-Chain Fatty Acids With Gut Microbiota and Clinical Severity in Patients With Parkinson Disease
Abstract
Background and objectives: Short-chain fatty acids (SCFAs) are gut microbial metabolites that promote the disease process in a rodent model of Parkinson disease (PD), but fecal levels of SCFAs in patients with PD are reduced. Simultaneous assessments of fecal and plasma SCFA levels, and their interrelationships with the PD disease process, are scarce. We aimed to compare fecal and plasma levels of different SCFA subtypes in patients with PD and healthy controls to delineate their interrelations and link to gut microbiota changes and clinical severity of PD.
Methods: A cohort of 96 patients with PD and 85 controls were recruited from National Taiwan University Hospital. Fecal and plasma concentrations of SCFAs were measured using chromatography and mass spectrometry. Gut microbiota was analyzed using metagenomic shotgun sequencing. Body mass index and medical comorbidities were evaluated and dietary information was obtained using a food frequency questionnaire. To assess motor and cognitive impairment, we used the Movement Disorder Society-Unified Parkinson's Disease Rating Scale (MDS-UPDRS) and the Mini-Mental Status Examination (MMSE).
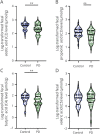
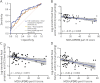
Results: Compared with controls, patients with PD had lower fecal but higher plasma concentrations of acetate, propionate, and butyrate. After adjustment for age, sex, disease duration, and anti-PD medication dosage, MDS-UPDRS part III motor scores correlated with reduced fecal levels of acetate (ρ = -0.37, p = 0.012), propionate (ρ = -0.32, p = 0.036), and butyrate (ρ = -0.40, p = 0.004) and with increased plasma propionate concentrations (ρ = 0.26, p = 0.042) in patients with PD. MMSE scores negatively correlated with plasma levels of butyrate (ρ = -0.09, p = 0.027) and valerate (ρ = -0.032, p = 0.033) after adjustment for confounders. SCFAs-producing gut bacteria correlated positively with fecal levels of SCFAs in healthy controls but revealed no association in patients with PD. In the PD patient group, the abundance of proinflammatory microbes, such as Clostridiales bacterium NK3B98 and Ruminococcus sp AM07-15, significantly correlated with decreased fecal levels and increased plasma levels of SCFAs, especially propionic acid.
Discussion: Reductions in fecal SCFAs but increased plasma SCFAs were observed in patients with PD and corelated to specific gut microbiota changes and the clinical severity of PD.
Classification of evidence: This study provides Class III evidence that gut metabolite SCFAs distinguish between patients with PD and controls and are associated with disease severity in patients with PD.
Copyright © 2022 The Author(s). Published by Wolters Kluwer Health, Inc. on behalf of the American Academy of Neurology.
Figures


Comment in
-
Author Response: Association of Fecal and Plasma Levels of Short-Chain Fatty Acids With Gut Microbiota and Clinical Severity in Patients With Parkinson Disease.Neurology. 2022 Aug 30;99(9):404. doi: 10.1212/WNL.0000000000201131. Neurology. 2022. PMID: 36038282 No abstract available.
-
Reader Response: Association of Fecal and Plasma Levels of Short-Chain Fatty Acids With Gut Microbiota and Clinical Severity in Patients With Parkinson Disease.Neurology. 2022 Aug 30;99(9):403. doi: 10.1212/WNL.0000000000201130. Neurology. 2022. PMID: 36038285 No abstract available.
References
-
- Klingelhoefer L, Reichmann H. Pathogenesis of Parkinson disease: the gut–brain axis and environmental factors. Nat Rev Neurol. 2015;11(11):625-636. - PubMed
-
- Schrag A, Horsfall L, Walters K, Noyce A, Petersen I. Prediagnostic presentations of Parkinson's disease in primary care: a case-control study. Lancet Neurol. 2015;14(1):57-64. - PubMed
-
- Nishiwaki H, Ito M, Ishida T, et al. Meta-analysis of gut dysbiosis in Parkinson's disease. Mov Disord. 2020;35(9):1626-1635. - PubMed
-
- Qian Y, Yang X, Xu S, et al. Gut metagenomics-derived genes as potential biomarkers of Parkinson's disease. Brain. 2020;143(8):2474-2489. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
