Blast-induced axonal degeneration in the rat cerebellum in the absence of head movement
- PMID: 34996954
- PMCID: PMC8741772
- DOI: 10.1038/s41598-021-03744-4
Blast-induced axonal degeneration in the rat cerebellum in the absence of head movement
Abstract
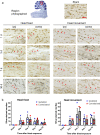
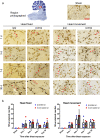
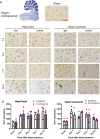
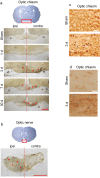
Blast exposure can injure brain by multiple mechanisms, and injury attributable to direct effects of the blast wave itself have been difficult to distinguish from that caused by rapid head displacement and other secondary processes. To resolve this issue, we used a rat model of blast exposure in which head movement was either strictly prevented or permitted in the lateral plane. Blast was found to produce axonal injury even with strict prevention of head movement. This axonal injury was restricted to the cerebellum, with the exception of injury in visual tracts secondary to ocular trauma. The cerebellar axonal injury was increased in rats in which blast-induced head movement was permitted, but the pattern of injury was unchanged. These findings support the contentions that blast per se, independent of head movement, is sufficient to induce axonal injury, and that axons in cerebellar white matter are particularly vulnerable to direct blast-induced injury.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
Rapid Repeat Exposure to Subthreshold Trauma Causes Synergistic Axonal Damage and Functional Deficits in the Visual Pathway in a Mouse Model.J Neurotrauma. 2019 May 15;36(10):1646-1654. doi: 10.1089/neu.2018.6046. Epub 2019 Jan 8. J Neurotrauma. 2019. PMID: 30451083 Free PMC article.
-
Model matters: Differential outcomes in traumatic optic neuropathy pathophysiology between blunt and blast-wave mediated head injuries.Exp Neurol. 2024 Feb;372:114613. doi: 10.1016/j.expneurol.2023.114613. Epub 2023 Nov 22. Exp Neurol. 2024. PMID: 37995952 Free PMC article.
-
Blast exposure in rats with body shielding is characterized primarily by diffuse axonal injury.J Neurotrauma. 2011 Jun;28(6):947-59. doi: 10.1089/neu.2010.1540. J Neurotrauma. 2011. PMID: 21449683
-
Traumatology of the optic nerve and contribution of crystallins to axonal regeneration.Cell Tissue Res. 2012 Jul;349(1):49-69. doi: 10.1007/s00441-012-1442-4. Epub 2012 May 26. Cell Tissue Res. 2012. PMID: 22638995 Review.
-
Visual system pathology in humans and animal models of blast injury.J Comp Neurol. 2017 Sep 1;525(13):2955-2967. doi: 10.1002/cne.24252. Epub 2017 Jun 20. J Comp Neurol. 2017. PMID: 28560719 Review.
Cited by
-
Neurological Effects of Repeated Blast Exposure in Special Operations Personnel.J Neurotrauma. 2024 Apr;41(7-8):942-956. doi: 10.1089/neu.2023.0309. Epub 2024 Jan 12. J Neurotrauma. 2024. PMID: 37950709 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

