Functional CDKN2A assay identifies frequent deleterious alleles misclassified as variants of uncertain significance
- PMID: 35001868
- PMCID: PMC8824478
- DOI: 10.7554/eLife.71137
Functional CDKN2A assay identifies frequent deleterious alleles misclassified as variants of uncertain significance
Abstract
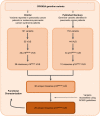
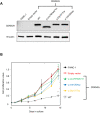
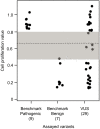
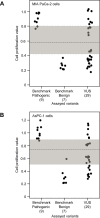
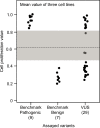
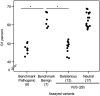
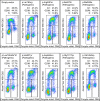
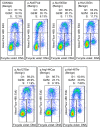
Pathogenic germline CDKN2A variants are associated with an increased risk of pancreatic ductal adenocarcinoma (PDAC). CDKN2A variants of uncertain significance (VUSs) are reported in up to 4.3% of patients with PDAC and result in significant uncertainty for patients and their family members as an unknown fraction are functionally deleterious, and therefore, likely pathogenic. Functional characterization of CDKN2A VUSs is needed to reclassify variants and inform clinical management. Twenty-nine germline CDKN2A VUSs previously reported in patients with PDAC or in ClinVar were evaluated using a validated in vitro cell proliferation assay. Twelve of the 29 CDKN2A VUSs were functionally deleterious (11 VUSs) or potentially functionally deleterious (1 VUS) and were reclassified as likely pathogenic variants. Thus, over 40% of CDKN2A VUSs identified in patients with PDAC are functionally deleterious and likely pathogenic. When incorporating VUSs found to be functionally deleterious, and reclassified as likely pathogenic, the prevalence of pathogenic/likely pathogenic CDKN2A in patients with PDAC reported in the published literature is increased to up to 4.1% of patients, depending on family history. Therefore, CDKN2A VUSs may play a significant, unappreciated role in risk of pancreatic cancer. These findings have significant implications for the counselling and care of patients and their relatives.
Keywords: CDKN2A; cancer; cancer biology; familial cancer; genetics; genomics; human; pancreas; pancreatic ductal adenocarcinoma; variant of uncertain significance.
© 2022, Kimura et al.
Conflict of interest statement
HK, NN, LW, JE, MG, AK, NR No competing interests declared, RH RHH has the right to receive royalty payments from Thrive Earlier Diagnosis for the GNAS in pancreatic cysts invention in a relationship overseen by Johns Hopkins University
Figures



References
-
- Bouvet D, Bodo S, Munier A, Guillerm E, Bertrand R, Colas C, Duval A, Coulet F, Muleris M. Methylation Tolerance-Based Functional Assay to Assess Variants of Unknown Significance in the MLH1 and MSH2 Genes and Identify Patients With Lynch Syndrome. Gastroenterology. 2019;157:421–431. doi: 10.1053/j.gastro.2019.03.071. - DOI - PubMed
-
- Brand R, Borazanci E, Speare V, Dudley B, Karloski E, Peters MLB, Stobie L, Bahary N, Zeh H, Zureikat A, Hogg M, Lee K, Tsung A, Rhee J, Ohr J, Sun W, Lee J, Moser AJ, DeLeonardis K, Krejdovsky J, Dalton E, LaDuca H, Dolinsky J, Colvin A, Lim C, Black MH, Tung N. Prospective study of germline genetic testing in incident cases of pancreatic adenocarcinoma. Cancer. 2018;124:3520–3527. doi: 10.1002/cncr.31628. - DOI - PubMed
-
- Canto MI, Harinck F, Hruban RH, Offerhaus GJ, Poley JW, Kamel I, Nio Y, Schulick RS, Bassi C, Kluijt I, Levy MJ, Chak A, Fockens P, Goggins M, Bruno M, International Cancer of Pancreas Screening CAPS Consortium International Cancer of the Pancreas Screening (CAPS) Consortium summit on the management of patients with increased risk for familial pancreatic cancer. Gut. 2013;62:339–347. doi: 10.1136/gutjnl-2012-303108. - DOI - PMC - PubMed
-
- Canto M.I, Almario JA, Schulick RD, Yeo CJ, Klein A, Blackford A, Shin EJ, Sanyal A, Yenokyan G, Lennon AM, Kamel IR, Fishman EK, Wolfgang C, Weiss M, Hruban RH, Goggins M. Risk of Neoplastic Progression in Individuals at High Risk for Pancreatic Cancer Undergoing Long-term Surveillance. Gastroenterology. 2018;155:740–751. doi: 10.1053/j.gastro.2018.05.035. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

