Lacrimal gland homeostasis is maintained by the AQP5 pathway by attenuating endoplasmic reticulum stress inflammation in the lacrimal gland of AQP5 knockout mice
- PMID: 35002213
- PMCID: PMC8684812
Lacrimal gland homeostasis is maintained by the AQP5 pathway by attenuating endoplasmic reticulum stress inflammation in the lacrimal gland of AQP5 knockout mice
Abstract
Purpose: AQP5-/- mice spontaneously exhibit dry eye symptoms. The purpose of this study was to assess the endoplasmic reticulum (ER) stress-mediated inflammation generated by a deficiency of aquaporin 5 (AQP5) in the lacrimal gland.
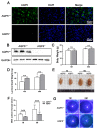
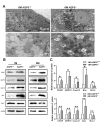
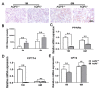
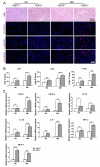
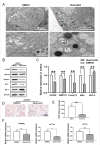
Methods: Hematoxylin and eosin (H&E) staining, Oil Red O staining, and transmission electron microscopy (TEM) analysis were performed to identify structural changes in lacrimal gland epithelial cells because of AQP5 deficiency. Corneal epithelial defects were assessed with sodium fluorescein staining. The expression profiles of mRNA and proteins were determined by quantitative real-time reverse transcription PCR (qRT-PCR) and western blot. Mice in the quercetin group were injected intraperitoneally with 40 mg/kg of quercetin, and the control group was injected with an equal volume of dimethyl sulfoxide (DMSO) for 4 weeks.
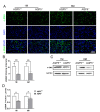
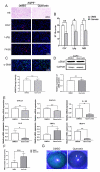
Results: Aqueous tear secretion fell at about 50% in 1- and 6-month-old AQP5-/- mice compared with that of AQP5+/+ mice. TEM showed that the ER structure was damaged. ER stress was significantly increased in the lacrimal gland of AQP5-/- mice. Lipid droplets accumulated in the matrix and acinar cells, and changes occurred in the lipid metabolism and gene expression levels for PPARα, CPT1α, and CPT2 in the AQP5-/- mice. Immune cell infiltration and increases in the gene expression levels of the chemokines CXCL1, CXCL2, and CCL5 were found in the lacrimal gland of AQP5-/- mice. Quercetin partially reversed ER stress levels, inflammation, and lipid accumulation, and it inhibited tear secretion.
Conclusions: The study data indicated that a deficiency of AQP5 induced pathophysiological changes and functional decompensation of the lacrimal gland. Quercetin may improve the inflammation in the lacrimal glands of AQP5-/- mice by regulating the ER stress levels.
Copyright © 2021 Molecular Vision.
Figures
Similar articles
-
Expression Profiles of CircRNA and mRNA in Lacrimal Glands of AQP5-/- Mice With Primary Dry Eye.Front Physiol. 2020 Sep 3;11:1010. doi: 10.3389/fphys.2020.01010. eCollection 2020. Front Physiol. 2020. PMID: 33013441 Free PMC article.
-
Acupuncture promotes tear secretion by up-regulating VIP/cAMP/PKA/AQP5 signaling in guinea pigs with aqueous tear deficiency dry eye.Zhen Ci Yan Jiu. 2023 Oct 25;48(10):1025-1032. doi: 10.13702/j.1000-0607.20230061. Zhen Ci Yan Jiu. 2023. PMID: 37879953 Chinese, English.
-
The difference of aquaporin 5 distribution in acinar and ductal cells in lacrimal and parotid glands.Curr Eye Res. 2007 Nov;32(11):923-9. doi: 10.1080/02713680701733076. Curr Eye Res. 2007. PMID: 18027168
-
Physiological role of aquaporin 5 in salivary glands.Pflugers Arch. 2016 Apr;468(4):519-39. doi: 10.1007/s00424-015-1749-6. Epub 2015 Nov 5. Pflugers Arch. 2016. PMID: 26537593 Review.
-
New insight into lacrimal gland function: Role of the duct epithelium in tear secretion.Ocul Surf. 2020 Oct;18(4):595-603. doi: 10.1016/j.jtos.2020.07.002. Epub 2020 Jul 21. Ocul Surf. 2020. PMID: 32707335 Review.
Cited by
-
The Role of Aquaporin 4 in Lacrimal Gland Ductal Fluid Secretion in Mice.Invest Ophthalmol Vis Sci. 2024 May 1;65(5):30. doi: 10.1167/iovs.65.5.30. Invest Ophthalmol Vis Sci. 2024. PMID: 38771571 Free PMC article.
-
Topical naltrexone increases aquaporin 5 production in the lacrimal gland and restores tear production in diabetic rats.Exp Biol Med (Maywood). 2024 May 2;249:10175. doi: 10.3389/ebm.2024.10175. eCollection 2024. Exp Biol Med (Maywood). 2024. PMID: 38756167 Free PMC article.
-
Histochemical Comparison of Human and Rat Lacrimal Glands: Implications for Bio-Engineering Studies.Transl Vis Sci Technol. 2022 Nov 1;11(11):10. doi: 10.1167/tvst.11.11.10. Transl Vis Sci Technol. 2022. PMID: 36374486 Free PMC article.
-
Exploring Immune Cell Diversity in the Lacrimal Glands of Healthy Mice: A Single-Cell RNA-Sequencing Atlas.Int J Mol Sci. 2024 Jan 19;25(2):1208. doi: 10.3390/ijms25021208. Int J Mol Sci. 2024. PMID: 38279208 Free PMC article.
-
Regulation of Axon Guidance by Slit2 and Netrin-1 Signaling in the Lacrimal Gland of Aqp5 Knockout Mice.Invest Ophthalmol Vis Sci. 2023 Sep 1;64(12):27. doi: 10.1167/iovs.64.12.27. Invest Ophthalmol Vis Sci. 2023. PMID: 37707834 Free PMC article.
References
-
- Bannier-Hélaouët M, Post Y, Korving J, Trani Bustos M, Gehart H, Begthel H, Bar-Ephraim YE, van der Vaart J, Kalmann R, Imhoff SM, Clevers H. Exploring the human lacrimal gland using organoids and single-cell sequencing. Cell Stem Cell. 2021;28:1221–32.e7. - PubMed
-
- Klećkowska-Nawrot J, Goździewska-Harłajczuk K, Kowalczyk A, Łukaszewicz E, Nowaczyk R. Histological, histochemical and ultrastructural studies on Harderian and lacrimal glands of the Capercaillie (Tetrao urogallus major L.). Acta Biol Hung. 2016;67:27–41. - PubMed
-
- Hodges RR, Dartt DA. Regulatory pathways in lacrimal gland epithelium. Int Rev Cytol. 2003;231:129–96. - PubMed
-
- Botelho SY. TEARS AND THE LACRIMAL GLAND. Sci Am. 1964;211:78–86. - PubMed
-
- Sullivan DA, Wickham LA, Rocha EM, Krenzer KL, Sullivan BD, Steagall R, Cermak JM, Dana MR, Ullman MD, Sato EH, Gao J, Rocha FJ, Ono M, Silveira LA, Lambert RW, Kelleher RS, Tolls DB, Toda I. Androgens and dry eye in Sjögren’s syndrome. Ann N Y Acad Sci. 1999;876:312–24. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

