Overview of follicle stimulating hormone and its receptors in reproduction and in stem cells and cancer stem cells
- PMID: 35002517
- PMCID: PMC8741861
- DOI: 10.7150/ijbs.63721
Overview of follicle stimulating hormone and its receptors in reproduction and in stem cells and cancer stem cells
Abstract
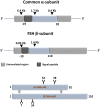
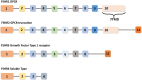
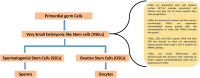
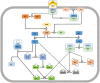
Follicle stimulating hormone (FSH) and its receptor (FSHR) have been reported to be responsible for several physiological functions and cancers. The responsiveness of stem cells and cancer stem cells towards the FSH-FSHR system make the function of FSH and its receptors more interesting in the context of cancer biology. This review is comprised of comprehensive information on FSH-FSHR signaling in normal physiology, gonadal stem cells, cancer cells, and potential options of utilizing FSH-FSHR system as an anti-cancer therapeutic target.
© The author(s).
Conflict of interest statement
Competing Interests: The authors have declared that no competing interest exists.
Figures



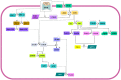

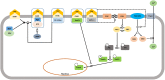
Similar articles
-
Follicle-Stimulating Hormone Receptor: Advances and Remaining Challenges.Int Rev Cell Mol Biol. 2018;338:1-58. doi: 10.1016/bs.ircmb.2018.02.001. Epub 2018 Apr 5. Int Rev Cell Mol Biol. 2018. PMID: 29699689 Review.
-
An overview of FSH-FSHR biology and explaining the existing conundrums.J Ovarian Res. 2021 Oct 30;14(1):144. doi: 10.1186/s13048-021-00880-3. J Ovarian Res. 2021. PMID: 34717708 Free PMC article. Review.
-
Differential FSH Glycosylation Modulates FSHR Oligomerization and Subsequent cAMP Signaling.Front Endocrinol (Lausanne). 2021 Dec 3;12:765727. doi: 10.3389/fendo.2021.765727. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 34925235 Free PMC article.
-
Evidence for Follicle-stimulating Hormone Receptor as a Functional Trimer.J Biol Chem. 2014 May 16;289(20):14273-82. doi: 10.1074/jbc.M114.549592. Epub 2014 Apr 1. J Biol Chem. 2014. PMID: 24692546 Free PMC article.
-
Follicle-Stimulating Hormone Induces Lipid Droplets via Gαi/o and β-Arrestin in an Endometrial Cancer Cell Line.Front Endocrinol (Lausanne). 2022 Feb 3;12:798866. doi: 10.3389/fendo.2021.798866. eCollection 2021. Front Endocrinol (Lausanne). 2022. PMID: 35185785 Free PMC article.
Cited by
-
Effects of follicle-stimulating hormone on the proliferation and apoptosis of infantile hemangioma stem cells.Biochem Biophys Rep. 2023 Sep 30;35:101551. doi: 10.1016/j.bbrep.2023.101551. eCollection 2023 Sep. Biochem Biophys Rep. 2023. PMID: 37823006 Free PMC article.
-
Comparison of mouse ovarian follicular development and gene expression in the presence of ovarian tissue extract and sodium selenite: An experimental study.Int J Reprod Biomed. 2023 May 12;21(5):415-424. doi: 10.18502/ijrm.v21i5.13476. eCollection 2023 May. Int J Reprod Biomed. 2023. PMID: 37362095 Free PMC article.
-
Significance of FSHR and LHCGR gene polymorphisms on clinical outcomes in gonadotropin-releasing hormone antagonist protocol with freeze-all strategy: A case-control study.Int J Reprod Biomed. 2024 Sep 12;22(7):539-552. doi: 10.18502/ijrm.v22i7.16962. eCollection 2024 Jul. Int J Reprod Biomed. 2024. PMID: 39355311 Free PMC article.
-
Effects of follicle-stimulating hormone on fat metabolism and cognitive impairment in women during menopause.Front Physiol. 2022 Dec 5;13:1043237. doi: 10.3389/fphys.2022.1043237. eCollection 2022. Front Physiol. 2022. PMID: 36545281 Free PMC article. Review.
-
Targeting the FSH/FSHR axis in ovarian cancer: advanced treatment using nanotechnology and immunotherapy.Front Endocrinol (Lausanne). 2024 Dec 17;15:1489767. doi: 10.3389/fendo.2024.1489767. eCollection 2024. Front Endocrinol (Lausanne). 2024. PMID: 39741875 Free PMC article. Review.
References
-
- Pierce JG, Parsons TF. Glycoprotein hormones: Structure and function. Annu Rev Biochem. 1981;50:465–95. - PubMed
-
- Richards JS. Maturation of ovarian follicles: actions and interactions of pituitary and ovarian hormones on follicular cell differentiation. Physiol Rev. 1980;60(1):51–89. - PubMed
-
- McLachlan RI, Wreford NG, Robertson DM, de Kretser DM. Hormonal control of spermatogenesis. Trends Endocrinol Metab. 1995;6(3):95–101. - PubMed
-
- Gharib SD, Chin WW, Wierman ME, Shupnik MA. Molecular biology of the pituitary gonadotropins. Endocr Rev. 1990;11(1):177–99. - PubMed
-
- Ulloa-Agirre A, Timossi C, Damian-Matsumara P, Dias JA. Role of Glycosylation on Folding, Assembly, and Secretion of hFSH. Endocrine. 1999;11(3):205–15. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

