Pro-Resolving Factors Released by Macrophages After Efferocytosis Promote Mucosal Wound Healing in Inflammatory Bowel Disease
- PMID: 35003066
- PMCID: PMC8727348
- DOI: 10.3389/fimmu.2021.754475
Pro-Resolving Factors Released by Macrophages After Efferocytosis Promote Mucosal Wound Healing in Inflammatory Bowel Disease
Abstract
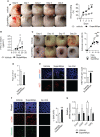
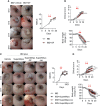
Nonresolving inflammation is a critical driver of several chronic inflammatory diseases, including inflammatory bowel diseases (IBD). This unresolved inflammation may result from the persistence of an initiating stimulus or from the alteration of the resolution phase of inflammation. Elimination of apoptotic cells by macrophages (a process called efferocytosis) is a critical step in the resolution phase of inflammation. Efferocytosis participates in macrophage reprogramming and favors the release of numerous pro-resolving factors. These pro-resolving factors exert therapeutic effects in experimental autoimmune arthritis. Here, we propose to evaluate the efficacy of pro-resolving factors produced by macrophages after efferocytosis, a secretome called SuperMApo, in two IBD models, namely dextran sodium sulfate (DSS)-induced and T cell transfer-induced colitis. Reintroducing these pro-resolving factors was sufficient to decrease clinical, endoscopic and histological colitis scores in ongoing naive T cell-transfer-induced colitis and in DSS-induced colitis. Mouse primary fibroblasts isolated from the colon demonstrated enhanced healing properties in the presence of SuperMApo, as attested by their increased migratory, proliferative and contractive properties. This was confirmed by the use of human fibroblasts isolated from patients with IBD. Exposure of an intestinal epithelial cell (IEC) line to these pro-resolving factors increased their proliferative properties and IEC acquired the capacity to capture apoptotic cells. The improvement of wound healing properties induced by SuperMApo was confirmed in vivo in a biopsy forceps-wound colonic mucosa model. Further in vivo analysis in naive T cell transfer-induced colitis model demonstrated an improvement of intestinal barrier permeability after administration of SuperMApo, an intestinal cell proliferation and an increase of α-SMA expression by fibroblasts, as well as a reduction of the transcript coding for fibronectin (Fn1). Finally, we identified TGF-β, IGF-I and VEGF among SuperMApo as necessary to favor mucosal healing and confirmed their role both in vitro (using neutralizing antibodies) and in vivo by depleting these factors from efferocytic macrophage secretome using antibody-coated microbeads. These growth factors only explained some of the beneficial effects induced by factors released by efferocytic macrophages. Overall, the administration of pro-resolving factors released by efferocytic macrophages limits intestinal inflammation and enhance tissue repair, which represents an innovative treatment of IBD.
Keywords: colitis; efferocytosis; fibroblasts; macrophages; resolution of inflammation; wound healing.
Copyright © 2021 Martin-Rodriguez, Gauthier, Bonnefoy, Couturier, Daoui, Chagué, Valmary-Degano, Gay, Saas and Perruche.
Conflict of interest statement
FB, MC, PS, and SP are shareholders of MED’INN’Pharma SAS. FB, MC, and SP are employed by MED’INN’Pharma SAS which develops the SuperMApo biologic drug candidate. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

