A review of advances in the last decade on targeted cancer therapy using 177Lu: focusing on 177Lu produced by the direct neutron activation route
- PMID: 35003885
- PMCID: PMC8727880
A review of advances in the last decade on targeted cancer therapy using 177Lu: focusing on 177Lu produced by the direct neutron activation route
Abstract
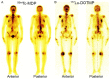
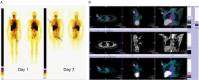
Lutetium-177 [T½ = 6.76 d; Eβ (max) = 0.497 MeV; maximum tissue range ~2.5 mm; 208 keV γ-ray] is one of the most important theranostic radioisotope used for the management of various oncological and non-oncological disorders. The present review chronicles the advancement in the last decade in 177Lu-radiopharmacy with a focus on 177Lu produced via direct 176Lu (n, γ) 177Lu nuclear reaction in medium flux research reactors. The specific nuances of 177Lu production by various routes are described and their pros and cons are discussed. Lutetium, is the last element in the lanthanide series. Its chemistry plays a vital role in the preparation of a wide variety of radiopharmaceuticals which demonstrate appreciable in vivo stability. Traditional bifunctional chelators (BFCs) that are used for 177Lu-labeling are discussed and the upcoming ones are highlighted. Research efforts that resulted in the growth of various 177Lu-based radiopharmaceuticals in preclinical and clinical settings are provided. This review also summarizes the results of clinical studies with potent 177Lu-based radiopharmaceuticals that have been prepared using medium specific activity 177Lu produced by direct neutron activation route in research reactors. Overall, the review amply demonstrates the practicality of the medium specific activity 177Lu towards formulation of various clinically useful radiopharmaceuticals, especially for the benefit of millions of cancer patients in developing countries with limited reactor facilities.
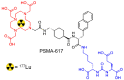
Keywords: 177Lu; DOTATATE; PSMA-617; TENIS; direct neutron activation; intrinsically radiolabeled nanoparticles; medium flux research reactors; specific activity; targeted therapy.
AJNMMI Copyright © 2021.
Conflict of interest statement
None.
Figures


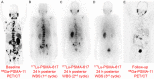




Similar articles
-
Safety and Therapeutic Optimization of Lutetium-177 Based Radiopharmaceuticals.Pharmaceutics. 2023 Apr 13;15(4):1240. doi: 10.3390/pharmaceutics15041240. Pharmaceutics. 2023. PMID: 37111725 Free PMC article. Review.
-
Therapeutic Multidose Preparation of a Ready-to-Use 177Lu-PSMA-617 Using Carrier Added Lutetium-177 in a Hospital Radiopharmacy and Its Clinical Efficacy.Cancer Biother Radiopharm. 2021 Oct;36(8):682-692. doi: 10.1089/cbr.2020.4261. Epub 2021 Aug 16. Cancer Biother Radiopharm. 2021. PMID: 34402687
-
Single vial kit formulation of DOTATATE for preparation of (177) Lu-labeled therapeutic radiopharmaceutical at hospital radiopharmacy.J Labelled Comp Radiopharm. 2015 Apr;58(4):166-72. doi: 10.1002/jlcr.3267. Epub 2015 Mar 12. J Labelled Comp Radiopharm. 2015. PMID: 25765604
-
Tracer level radiochemistry to clinical dose preparation of (177)Lu-labeled cyclic RGD peptide dimer.Nucl Med Biol. 2013 Oct;40(7):946-54. doi: 10.1016/j.nucmedbio.2013.05.011. Epub 2013 Jul 11. Nucl Med Biol. 2013. PMID: 23850487
-
Production of (177)Lu for Targeted Radionuclide Therapy: Available Options.Nucl Med Mol Imaging. 2015 Jun;49(2):85-107. doi: 10.1007/s13139-014-0315-z. Epub 2015 Feb 17. Nucl Med Mol Imaging. 2015. PMID: 26085854 Free PMC article. Review.
Cited by
-
Safety and Therapeutic Optimization of Lutetium-177 Based Radiopharmaceuticals.Pharmaceutics. 2023 Apr 13;15(4):1240. doi: 10.3390/pharmaceutics15041240. Pharmaceutics. 2023. PMID: 37111725 Free PMC article. Review.
-
Nanotechnology-Assisted Cell Tracking.Nanomaterials (Basel). 2022 Apr 20;12(9):1414. doi: 10.3390/nano12091414. Nanomaterials (Basel). 2022. PMID: 35564123 Free PMC article. Review.
-
Enhancing the radionuclide theranostic concept through the radiohybrid approach.RSC Med Chem. 2024 Nov 25;16(5):1856-1864. doi: 10.1039/d4md00591k. eCollection 2025 May 22. RSC Med Chem. 2024. PMID: 39618962 Free PMC article. Review.
-
Cancer Brachytherapy at the Nanoscale: An Emerging Paradigm.Chem Biomed Imaging. 2023 Nov 21;2(1):4-26. doi: 10.1021/cbmi.3c00092. eCollection 2024 Jan 22. Chem Biomed Imaging. 2023. PMID: 38274040 Free PMC article. Review.
-
Optimization of 177Lu-labelling of DOTA-TOC, PSMA-I&T and FAPI-46 for clinical application.EJNMMI Radiopharm Chem. 2023 May 26;8(1):10. doi: 10.1186/s41181-023-00196-1. EJNMMI Radiopharm Chem. 2023. PMID: 37233924 Free PMC article.
References
-
- Choudhury P, Gupta M. Personalized & precision medicine in cancer: a theranostic approach. Curr Radiopharm. 2017;10:166–170. - PubMed
-
- Farolfi A, Lima GM, Oyen W, Fanti S. Molecular imaging and theranostics-a multidisciplinary approach. Semin Nucl Med. 2019;49:247–254. - PubMed
-
- Filippi L, Chiaravalloti A, Schillaci O, Cianni R, Bagni O. Theranostic approaches in nuclear medicine: current status and future prospects. Expert Rev Med Devices. 2020;17:331–343. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
