Degradation of Components of the Lpt Transenvelope Machinery Reveals LPS-Dependent Lpt Complex Stability in Escherichia coli
- PMID: 35004843
- PMCID: PMC8727689
- DOI: 10.3389/fmolb.2021.758228
Degradation of Components of the Lpt Transenvelope Machinery Reveals LPS-Dependent Lpt Complex Stability in Escherichia coli
Abstract
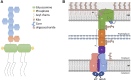
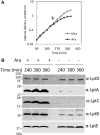
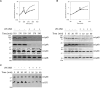
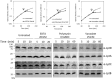
Lipopolysaccharide (LPS) is a peculiar component of the outer membrane (OM) of many Gram-negative bacteria that renders these bacteria highly impermeable to many toxic molecules, including antibiotics. LPS is assembled at the OM by a dedicated intermembrane transport system, the Lpt (LPS transport) machinery, composed of seven essential proteins located in the inner membrane (IM) (LptB2CFG), periplasm (LptA), and OM (LptDE). Defects in LPS transport compromise LPS insertion and assembly at the OM and result in an overall modification of the cell envelope and its permeability barrier properties. LptA is a key component of the Lpt machine. It connects the IM and OM sub-complexes by interacting with the IM protein LptC and the OM protein LptD, thus enabling the LPS transport across the periplasm. Defects in Lpt system assembly result in LptA degradation whose stability can be considered a marker of an improperly assembled Lpt system. Indeed, LptA recruitment by its IM and OM docking sites requires correct maturation of the LptB2CFG and LptDE sub-complexes, respectively. These quality control checkpoints are crucial to avoid LPS mistargeting. To further dissect the requirements for the complete Lpt transenvelope bridge assembly, we explored the importance of LPS presence by blocking its synthesis using an inhibitor compound. Here, we found that the interruption of LPS synthesis results in the degradation of both LptA and LptD, suggesting that, in the absence of the LPS substrate, the stability of the Lpt complex is compromised. Under these conditions, DegP, a major chaperone-protease in Escherichia coli, is responsible for LptD but not LptA degradation. Importantly, LptD and LptA stability is not affected by stressors disturbing the integrity of LPS or peptidoglycan layers, further supporting the notion that the LPS substrate is fundamental to keeping the Lpt transenvelope complex assembled and that LptA and LptD play a major role in the stability of the Lpt system.
Keywords: Lpt system; LpxC inhibitor; bacterial cell envelope; lipopolysaccharide; outer membrane stability.
Copyright © 2021 Martorana, Moura, Sperandeo, Di Vincenzo, Liang, Toone, Zhou and Polissi.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
New insights into the Lpt machinery for lipopolysaccharide transport to the cell surface: LptA-LptC interaction and LptA stability as sensors of a properly assembled transenvelope complex.J Bacteriol. 2011 Mar;193(5):1042-53. doi: 10.1128/JB.01037-10. Epub 2010 Dec 17. J Bacteriol. 2011. PMID: 21169485 Free PMC article.
-
Functional Interaction between the Cytoplasmic ABC Protein LptB and the Inner Membrane LptC Protein, Components of the Lipopolysaccharide Transport Machinery in Escherichia coli.J Bacteriol. 2016 Jul 28;198(16):2192-203. doi: 10.1128/JB.00329-16. Print 2016 Aug 15. J Bacteriol. 2016. PMID: 27246575 Free PMC article.
-
Thanatin Impairs Lipopolysaccharide Transport Complex Assembly by Targeting LptC-LptA Interaction and Decreasing LptA Stability.Front Microbiol. 2020 May 13;11:909. doi: 10.3389/fmicb.2020.00909. eCollection 2020. Front Microbiol. 2020. PMID: 32477309 Free PMC article.
-
Structural Insights into the Lipopolysaccharide Transport (Lpt) System as a Novel Antibiotic Target.J Microbiol. 2024 Apr;62(4):261-275. doi: 10.1007/s12275-024-00137-w. Epub 2024 May 31. J Microbiol. 2024. PMID: 38816673 Review.
-
Structural insight into lipopolysaccharide transport from the Gram-negative bacterial inner membrane to the outer membrane.Biochim Biophys Acta Mol Cell Biol Lipids. 2017 Nov;1862(11):1461-1467. doi: 10.1016/j.bbalip.2017.08.003. Epub 2017 Aug 15. Biochim Biophys Acta Mol Cell Biol Lipids. 2017. PMID: 28821406 Review.
Cited by
-
Suppressor Mutations in LptF Bypass Essentiality of LptC by Forming a Six-Protein Transenvelope Bridge That Efficiently Transports Lipopolysaccharide.mBio. 2023 Feb 28;14(1):e0220222. doi: 10.1128/mbio.02202-22. Epub 2022 Dec 21. mBio. 2023. PMID: 36541759 Free PMC article.
-
Determination of Initial Rates of Lipopolysaccharide Transport.Biochemistry. 2024 Oct 1;63(19):2440-2448. doi: 10.1021/acs.biochem.4c00379. Epub 2024 Sep 12. Biochemistry. 2024. PMID: 39264328 Free PMC article.
-
Thanatin: A Promising Antimicrobial Peptide Targeting the Achilles' Heel of Multidrug-Resistant Bacteria.Int J Mol Sci. 2024 Aug 31;25(17):9496. doi: 10.3390/ijms25179496. Int J Mol Sci. 2024. PMID: 39273441 Free PMC article. Review.
-
Making a chink in their armor: Current and next-generation antimicrobial strategies against the bacterial cell envelope.Adv Microb Physiol. 2023;83:221-307. doi: 10.1016/bs.ampbs.2023.05.003. Epub 2023 Jun 27. Adv Microb Physiol. 2023. PMID: 37507160 Free PMC article.
-
The transmembrane α-helix of LptC participates in LPS extraction by the LptB2 FGC transporter.Mol Microbiol. 2022 Jul;118(1-2):61-76. doi: 10.1111/mmi.14952. Epub 2022 Jun 27. Mol Microbiol. 2022. PMID: 35678757 Free PMC article.
References
-
- Benedet M., Falchi F. A., Puccio S., Di Benedetto C., Peano C., Polissi A., et al. (2006). The Lack of the Essential LptC Protein in the Trans-envelope Lipopolysaccharide Transport Machine Is Circumvented by Suppressor Mutations in LptF, an Inner Membrane Component of the Escherichia coli Transporter. PLoS One 11 (8), e0161354. 10.1371/journal.pone.0161354 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

