South Asian-Specific MYBPC3 Δ25bp Deletion Carriers Display Hypercontraction and Impaired Diastolic Function Under Exercise Stress
- PMID: 35004883
- PMCID: PMC8733148
- DOI: 10.3389/fcvm.2021.766339
South Asian-Specific MYBPC3 Δ25bp Deletion Carriers Display Hypercontraction and Impaired Diastolic Function Under Exercise Stress
Abstract
Background: A 25-base pair (25bp) intronic deletion in the MYBPC3 gene enriched in South Asians (SAs) is a risk allele for late-onset left ventricular (LV) dysfunction, hypertrophy, and heart failure (HF) with several forms of cardiomyopathy. However, the effect of this variant on exercise parameters has not been evaluated. Methods: As a pilot study, 10 asymptomatic SA carriers of the MYBPC3 Δ25bp variant (52.9 ± 2.14 years) and 10 age- and gender-matched non-carriers (NCs) (50.1 ± 2.7 years) were evaluated at baseline and under exercise stress conditions using bicycle exercise echocardiography and continuous cardiac monitoring. Results: Baseline echocardiography parameters were not different between the two groups. However, in response to exercise stress, the carriers of Δ25bp had significantly higher LV ejection fraction (%) (CI: 4.57 ± 1.93; p < 0.0001), LV outflow tract peak velocity (m/s) (CI: 0.19 ± 0.07; p < 0.0001), and higher aortic valve (AV) peak velocity (m/s) (CI: 0.103 ± 0.08; p = 0.01) in comparison to NCs, and E/A ratio, a marker of diastolic compliance, was significantly lower in Δ25bp carriers (CI: 0.107 ± 0.102; p = 0.038). Interestingly, LV end-diastolic diameter (LVIDdia) was augmented in NCs in response to stress, while it did not increase in Δ25bp carriers (CI: 0.239 ± 0.125; p = 0.0002). Further, stress-induced right ventricular systolic excursion velocity s' (m/s), as a marker of right ventricle function, increased similarly in both groups, but tricuspid annular plane systolic excursion increased more in carriers (slope: 0.008; p = 0.0001), suggesting right ventricle functional differences between the two groups. Conclusions: These data support that MYBPC3 Δ25bp is associated with LV hypercontraction under stress conditions with evidence of diastolic impairment.
Keywords: 25bp deletion; DOSA study; MYBPC3; South Asians; hypertrophic cardiomyopathy; myosin binding protein-C; stress echocardiography; ventricular diastolic dysfunction.
Copyright © 2021 Bazrafshan, Sibilia, Girgla, Viswanathan, Puckelwartz, Sangha, Singh, Kakroo, Jandarov, Harris, Rubinstein, Becker, McNally and Sadayappan.
Conflict of interest statement
SS provides consulting and collaborative research studies to the Leducq Foundation (CURE-PLAN), Red Saree Inc., Greater Cincinnati Tamil Sangam, Pfizer, Novo Nordisk, AstraZeneca, MyoKardia, Merck, and Amgen. EM serves as a consultant to AstraZeneca, Amgen, Pfizer, Tenaya Therapeutics, and Invitae. RB serves on scientific advisory boards for Janssen and Basking Biosciences and DSMB Committees for Ionis Pharmaceuticals, Akcea Therapeutics, and Novartis. These activities are unrelated to the content of this work. RRS has been a postdoctoral fellow of Amgen, starting from June 2019, and performs research at the University of Cincinnati. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


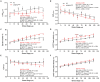
References
-
- Maron BJ, Gardin JM, Flack JM, Gidding SS, Kurosaki TT, Bild DE. Prevalence of hypertrophic cardiomyopathy in a general population of young adults. Echocardiographic analysis of 4111 subjects in the CARDIA Study Coronary Artery Risk Development in (Young) Adults. Circulation. (1995) 92:785–9. 10.1161/01.CIR.92.4.785 - DOI - PubMed
-
- Ommen SR, Mital S, Burke MA, Day SM, Deswal A, Elliott P, et al. . 2020 AHA/ACC Guideline for the diagnosis and treatment of patients with hypertrophic cardiomyopathy: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. (2020) 142:e533–57. 10.1161/CIR.0000000000000938 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

