Magnolol protects against acute gastrointestinal injury in sepsis by down-regulating regulated on activation, normal T-cell expressed and secreted
- PMID: 35004977
- PMCID: PMC8686136
- DOI: 10.12998/wjcc.v9.i34.10451
Magnolol protects against acute gastrointestinal injury in sepsis by down-regulating regulated on activation, normal T-cell expressed and secreted
Abstract
Background: Sepsis is a major medical challenge. Magnolol is an active constituent of Houpu that improves tissue function and exerts strong anti-endotoxin and anti-inflammatory effects, but the mechanism by which it reduces intestinal inflammation in sepsis is yet unclear.
Aim: To assess the protective effect of magnolol on intestinal mucosal epithelial cells in sepsis and elucidate the underlying mechanisms.
Methods: Enzyme-linked immunosorbent assay was used to measure tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), IL-6, and regulated on activation, normal T-cell expressed and secreted (RANTES) levels in serum and ileal tissue in animal studies. The histopathological changes of the ileal mucosa in different groups were observed under a microscope. Cell Counting Kit-8 and cell permeability assays were used to determine the concentration of drug-containing serum that did not affect the activity of Caco2 cells but inhibited lipopolysaccharide (LPS)-induced decrease in permeability. Immunofluorescence and Western blot assays were used to detect the levels of RANTES, inhibitor of nuclear factor kappa-B kinase β (IKKβ), phosphorylated IKKβ (p-IKKβ), inhibitor of nuclear factor kappa-B kinase α (IκBα), p65, and p-p65 proteins in different groups in vitro.
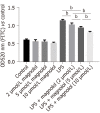
Results: In rats treated with LPS by intravenous tail injection in the presence or absence of magnolol, magnolol inhibited the expression of proinflammatory cytokines, IL-1β, IL-6, and TNF-α in a dose-dependent manner. In addition, magnolol suppressed the production of RANTES in LPS-stimulated sepsis rats. Moreover, in vitro studies suggested that magnolol inhibited the increase of p65 nucleation, thereby markedly downregulating the production of the phosphorylated form of IKKβ in LPS-treated Caco2 cells. Specifically, magnolol inhibited the translocation of the transcription factor nuclear factor-kappa B (NF-κB) from the cytosol into the nucleus and down-regulated the expression level of the chemokine RANTES in LPS-stimulated Caco2 cells.
Conclusion: Magnolol down-regulates RANTES levels by inhibiting the LPS/NF-κB signaling pathways, thereby suppressing IL-1β, IL-6, and TNF-α expression to alleviate the mucosal barrier dysfunction in sepsis.
Keywords: Anti-inflammation; Lipopolysaccharide; Magnolol; Nuclear factor-kappa B; Regulated on activation, normal T-cell expressed and secreted; Sepsis.
©The Author(s) 2021. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: We declare that we have no financial and personal relationships with other people or organizations that can inappropriately influence our work, and there is no professional or other personal interest of any nature or kind in any product, service and/or company that could be construed as influencing the position presented in the manuscript entitled.
Figures



References
-
- Levy MM, Evans LE, Rhodes A. The Surviving Sepsis Campaign Bundle: 2018 update. Intensive Care Med. 2018;44:925–928. - PubMed
-
- Perner A, Cecconi M, Cronhjort M, Darmon M, Jakob SM, Pettilä V, van der Horst ICC. Expert statement for the management of hypovolemia in sepsis. Intensive Care Med. 2018;44:791–798. - PubMed
-
- Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, Bellomo R, Bernard GR, Chiche JD, Coopersmith CM, Hotchkiss RS, Levy MM, Marshall JC, Martin GS, Opal SM, Rubenfeld GD, van der Poll T, Vincent JL, Angus DC. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3) JAMA. 2016;315:801–810. - PMC - PubMed
-
- Reintam Blaser A, Malbrain ML, Starkopf J, Fruhwald S, Jakob SM, De Waele J, Braun JP, Poeze M, Spies C. Gastrointestinal function in intensive care patients: terminology, definitions and management. Recommendations of the ESICM Working Group on Abdominal Problems. Intensive Care Med. 2012;38:384–394. - PMC - PubMed
-
- Gao Y, Han F, Huang X, Rong Y, Yi H, Wang Y. Changes in gut microbial populations, intestinal morphology, expression of tight junction proteins, and cytokine production between two pig breeds after challenge with Escherichia coli K88: a comparative study. J Anim Sci. 2013;91:5614–5625. - PubMed
LinkOut - more resources
Full Text Sources

