Native extracellular matrix orientation determines multipotent vascular stem cell proliferation in response to cyclic uniaxial tensile strain and simulated stent indentation
- PMID: 35005255
- PMCID: PMC8715293
- DOI: 10.1016/j.bbrep.2021.101183
Native extracellular matrix orientation determines multipotent vascular stem cell proliferation in response to cyclic uniaxial tensile strain and simulated stent indentation
Abstract
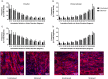
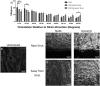
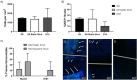
Cardiovascular disease is the leading cause of death worldwide, with multipotent vascular stem cells (MVSC) implicated in contributing to diseased vessels. MVSC are mechanosensitive cells which align perpendicular to cyclic uniaxial tensile strain. Within the blood vessel wall, collagen fibers constrain cells so that they are forced to align circumferentially, in the primary direction of tensile strain. In these experiments, MVSC were seeded onto the medial layer of decellularized porcine carotid arteries, then exposed to 10%, 1 Hz cyclic tensile strain for 10 days with the collagen fiber direction either parallel or perpendicular to the direction of strain. Cells aligned with the direction of the collagen fibers regardless of the orientation to strain. Cells aligned with the direction of strain showed an increased number of proliferative Ki67 positive cells, while those strained perpendicular to the direction of cell alignment showed no change in cell proliferation. A bioreactor system was designed to simulate the indentation of a single, wire stent strut. After 10 days of cyclic loading to 10% strain, MVSC showed regions of densely packed, highly proliferative cells. Therefore, MVSC may play a significant role in in-stent restenosis, and this proliferative response could potentially be controlled by controlling MVSC orientation relative to applied strain.
Keywords: Collagen; Cyclic tensile strain; ECM, Extracellular Matrix; Extracellular matrix; In-stent restenosis; MVSC, Multipotent vascular stem cell; Multipotent vascular stem cell; Proliferation; VSMC, Vascular smooth muscle cell.
© 2021 The Authors.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures

References
-
- Benjamin E.J., Muntner P., Alonso A., Bittencourt M.S., Callaway C.W., Carson A.P., Chamberlain A.M., Chang A.R., Cheng S., Das S.R., Delling F.N., Djousse L., Elkind M.S.V., Ferguson J.F., Fornage M., Jordan L.C., Khan S.S., Kissela B.M., Knutson K.L., Kwan T.W., Lackland D.T., Lewis T.T., Lichtman J.H., Longenecker C.T., Loop M.S., Lutsey P.L., Martin S.S., Matsushita K., Moran A.E., Mussolino M.E., O'Flaherty M., Pandey A., Perak A.M., Rosamond W.D., Roth G.A., Sampson U.K.A., Satou G.M., Schroeder E.B., Shah S.H., Spartano N.L., Stokes A., Tirschwell D.L., Tsao C.W., Turakhia M.P., VanWagner L.B., Wilkins J.T., Wong S.S., Virani S.S. Heart disease and stroke statistics-2019 update: a report from the American heart association. Circulation. 2019;139:e56–e528. doi: 10.1161/cir.0000000000000659. - DOI - PubMed
-
- Newby A.C. An overview of the vascular response to injury: a tribute to the late Russell Ross. Toxicol. Lett. 2000;112–113:519–529. - PubMed
LinkOut - more resources
Full Text Sources

