Venetoclax for Children and Adolescents with Acute Lymphoblastic Leukemia and Lymphoblastic Lymphoma
- PMID: 35008312
- PMCID: PMC8750927
- DOI: 10.3390/cancers14010150
Venetoclax for Children and Adolescents with Acute Lymphoblastic Leukemia and Lymphoblastic Lymphoma
Abstract
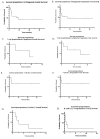
Venetoclax is approved for adult patients with chronic lymphocytic leukemia and acute myeloid leukemia. Expanding its use to the pediatric population is currently under investigation, but more robust data are needed. We retrospectively analyzed the safety and efficacy of venetoclax in children/AYA with ALL/LBL. We identified 18 patients (T-cell ALL, n = 7; T-cell LBL, n = 6; B-cell ALL, n = 5) aged 6-22 years. No new venetoclax safety signals were identified; the most common toxicity was myelosuppression. No deaths occurred within 30 days from the start of the therapy. A mean of 2.6 (range 0-8) prior lines of therapy were given. The mean duration of venetoclax was 4.06 months (range 0.2-24.67 months). Complete remission was achieved in 11 (61%) patients. Of the eight patients who remain alive, four are continuing on venetoclax combination therapy, and four proceeded to hematopoietic stem cell transplantation. Three patients who initially achieved CR, later relapsed, and are deceased. Nine patients are deceased, and one patient was lost to follow-up. Overall survival is 9.14 months (range 1.1-33.1), and progression-free survival is 7.34 months (range 0.2-33.1). This is the largest cohort of pediatric/AYA patients who received venetoclax for ALL/LBL. Our data support the consideration of venetoclax-based regimens in pediatric patients with R/R ALL/LBL and its investigation as upfront therapy for T-cell ALL/LBL.
Keywords: Bcl-2 inhibitor; acute lymphoblastic leukemia; early precursor T-cell; lymphoblastic lymphoma; venetoclax.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Gaynon P.S., Angiolillo A.L., Carroll W.L., Nachman J.B., Trigg M.E., Sather H.N., Hunger S.P., Devidas M. Long-term results of the children’s cancer group studies for childhood acute lymphoblastic leukemia 1983–2002: A Children’s Oncology Group Report. Leukemia. 2010;24:285–297. doi: 10.1038/leu.2009.262. - DOI - PMC - PubMed
-
- Salzer W.L., Devidas M., Carroll W.L., Winick N., Pullen J., Hunger S.P., Camitta B.A. Long-term results of the pediatric oncology group studies for childhood acute lymphoblastic leukemia 1984–2001: A report from the children’s oncology group. Leukemia. 2010;24:355–370. doi: 10.1038/leu.2009.261. - DOI - PMC - PubMed
-
- Möricke A., Zimmermann M., Reiter A., Henze G., Schrauder A., Gadner H., Ludwig W.D., Ritter J., Harbott J., Mann G., et al. Long-term results of five consecutive trials in childhood acute lymphoblastic leukemia performed by the ALL-BFM study group from 1981 to 2000. Leukemia. 2010;24:265–284. doi: 10.1038/leu.2009.257. - DOI - PubMed
-
- Silverman L.B., Stevenson K.E., O’Brien J.E., Asselin B.L., Barr R.D., Clavell L., Cole P.D., Kelly K.M., Laverdiere C., Michon B., et al. Long-term results of Dana-Farber Cancer Institute ALL Consortium protocols for children with newly diagnosed acute lymphoblastic leukemia (1985–2000) Leukemia. 2010;24:320–334. doi: 10.1038/leu.2009.253. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Medical

