Effect of Everolimus versus Bone Marrow-Derived Stem Cells on Glomerular Injury in a Rat Model of Glomerulonephritis: A Preventive, Predictive and Personalized Implication
- PMID: 35008770
- PMCID: PMC8745690
- DOI: 10.3390/ijms23010344
Effect of Everolimus versus Bone Marrow-Derived Stem Cells on Glomerular Injury in a Rat Model of Glomerulonephritis: A Preventive, Predictive and Personalized Implication
Abstract
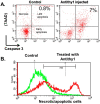
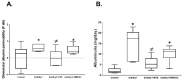
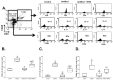
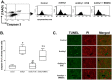
Glomerular endothelial injury and effectiveness of glomerular endothelial repair play a crucial role in the progression of glomerulonephritis. Although the potent immune suppressive everolimus is increasingly used in renal transplant patients, adverse effects of its chronic use have been reported clinically in human glomerulonephritis and experimental renal disease. Recent studies suggest that progenitor stem cells could enhance glomerular endothelial repair with minimal adverse effects. Increasing evidence supports the notion that stem cell therapy and regenerative medicine can be effectively used in pathological conditions within the predictive, preventive and personalized medicine (PPPM) paradigm. In this study, using an experimental model of glomerulonephritis, we tested whether bone marrow-derived stem cells (BMDSCs) could provide better effect over everolimus in attenuating glomerular injury and improving the repair process in a rat model of glomerulonephritis. Anti-Thy1 glomerulonephritis was induced in male Sprague Dawley rats by injection of an antibody against Thy1, which is mainly expressed on glomerular mesangial cells. Additional groups of rats were treated with the immunosuppressant everolimus daily after the injection of anti-Thy1 or injected with single bolus dose of BMDSCs after one week of injection of anti-Thy1 (n = 6-8). Nine days after injection of anti-Thy1, glomerular albumin permeability and albuminuria were significantly increased when compared to control group (p < 0.05). Compared to BMDSCs, everolimus was significantly effective in attenuating glomerular injury, nephrinuria and podocalyxin excretion levels as well as in reducing inflammatory responses and apoptosis. Our findings suggest that bolus injection of BMDSCs fails to improve glomerular injury whereas everolimus slows the progression of glomerular injury in Anti-Thy-1 induced glomerulonephritis. Thus, everolimus could be used at the early stage of glomerulonephritis, suggesting potential implications of PPPM in the treatment of progressive renal injury.
Keywords: BMDSCs; anti-Thy1; apoptosis; everolimus; glomerular injury; renal inflammation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Naik R.H., Shawar S.H. Rapidly Progressive Glomerulonephritis. StatPearls; Treasure Island, FL, USA: 2021.
-
- Rampino T., Gregorini M., Bedino G., Piotti G., Gabanti E., Ibatici A., Sessarego N., Piacenza C., Balenzano C.T., Esposito P., et al. Mesenchymal stromal cells improve renal injury in anti-Thy 1 nephritis by modulating inflammatory cytokines and scatter factors. Clin. Sci. 2010;120:25–36. doi: 10.1042/CS20100147. - DOI - PubMed
-
- Jefferson J.A., Johnson R.J. Experimental mesangial proliferative glomerulonephritis (the anti-Thy-1.1 model) J. Nephrol. 2000;12:297–307. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

