Cancer Therapy-Induced Cardiotoxicity-A Metabolic Perspective on Pathogenesis, Diagnosis and Therapy
- PMID: 35008867
- PMCID: PMC8745714
- DOI: 10.3390/ijms23010441
Cancer Therapy-Induced Cardiotoxicity-A Metabolic Perspective on Pathogenesis, Diagnosis and Therapy
Abstract
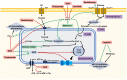
Long-term cardiovascular complications of cancer therapy are becoming ever more prevalent due to increased numbers of cancer survivors. Cancer therapy-induced cardiotoxicity (CTIC) is an incompletely understood consequence of various chemotherapies, targeted anti-cancer agents and radiation therapy. It is typically detected clinically by a reduction in cardiac left ventricular ejection fraction, assessed by echocardiography. However, once cardiac functional decline is apparent, this indicates irreversible cardiac damage, highlighting a need for the development of diagnostics which can detect CTIC prior to the onset of functional decline. There is increasing evidence to suggest that pathological alterations to cardiac metabolism play a crucial role in the development of CTIC. This review discusses the metabolic alterations and mechanisms which occur in the development of CTIC, with a focus on doxorubicin, trastuzumab, imatinib, ponatinib, sunitinib and radiotherapy. Potential methods to diagnose and predict CTIC prior to functional cardiac decline in the clinic are evaluated, with a view to both biomarker and imaging-based approaches. Finally, the therapeutic potential of therapies which manipulate cardiac metabolism in the context of adjuvant cardioprotection against CTIC is examined. Together, an integrated view of the role of metabolism in pathogenesis, diagnosis and treatment is presented.
Keywords: cardioprotection; cardiotoxicity; chemotherapy; heart failure; metabolism.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Cardinale D., Colombo A., Sandri M.T., Lamantia G., Colombo N., Civelli M., Martinelli G., Veglia F., Fiorentini C., Cipolla C.M. Prevention of high-dose chemotherapy-induced cardiotoxicity in high-risk patients by angiotensin-converting enzyme inhibition. Circulation. 2006;114:2474–2481. doi: 10.1161/CIRCULATIONAHA.106.635144. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

