Graphene-Oxide Porous Biopolymer Hybrids Enhance In Vitro Osteogenic Differentiation and Promote Ectopic Osteogenesis In Vivo
- PMID: 35008918
- PMCID: PMC8745160
- DOI: 10.3390/ijms23010491
Graphene-Oxide Porous Biopolymer Hybrids Enhance In Vitro Osteogenic Differentiation and Promote Ectopic Osteogenesis In Vivo
Abstract
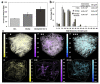
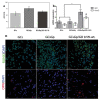
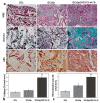
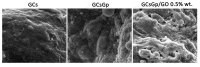
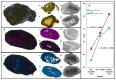
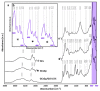
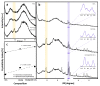
Over the years, natural-based scaffolds have presented impressive results for bone tissue engineering (BTE) application. Further, outstanding interactions have been observed during the interaction of graphene oxide (GO)-reinforced biomaterials with both specific cell cultures and injured bone during in vivo experimental conditions. This research hereby addresses the potential of fish gelatin/chitosan (GCs) hybrids reinforced with GO to support in vitro osteogenic differentiation and, further, to investigate its behavior when implanted ectopically. Standard GCs formulation was referenced against genipin (Gp) crosslinked blend and 0.5 wt.% additivated GO composite (GCsGp/GO 0.5 wt.%). Pre-osteoblasts were put in contact with these composites and induced to differentiate in vitro towards mature osteoblasts for 28 days. Specific bone makers were investigated by qPCR and immunolabeling. Next, CD1 mice models were used to assess de novo osteogenic potential by ectopic implantation in the subcutaneous dorsum pocket of the animals. After 4 weeks, alkaline phosphate (ALP) and calcium deposits together with collagen synthesis were investigated by biochemical analysis and histology, respectively. Further, ex vivo materials were studied after surgery regarding biomineralization and morphological changes by means of qualitative and quantitative methods. Furthermore, X-ray diffraction and Fourier-transform infrared spectroscopy underlined the newly fashioned material structuration by virtue of mineralized extracellular matrix. Specific bone markers determination stressed the osteogenic phenotype of the cells populating the material in vitro and successfully differentiated towards mature bone cells. In vivo results of specific histological staining assays highlighted collagen formation and calcium deposits, which were further validated by micro-CT. It was observed that the addition of 0.5 wt.% GO had an overall significant positive effect on both in vitro differentiation and in vivo bone cell recruitment in the subcutaneous region. These data support the GO bioactivity in osteogenesis mechanisms as being self-sufficient to elevate osteoblast differentiation and bone formation in ectopic sites while lacking the most common osteoinductive agents.
Keywords: biomineralization; biopolymer blends; ectopic bone formation; ex vivo analysis; graphene oxide; osteoinduction.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Foroutan S., Hashemian M., Khosravi M., Nejad M.G., Asefnejad A., Saber-Samandari S., Khandan A. A Porous Sodium Alginate-CaSiO 3 Polymer Reinforced with Graphene Nanosheet: Fabrication and Optimality Analysis. Fibers Polym. 2021;22:540–549. doi: 10.1007/s12221-021-0347-9. - DOI
-
- Di Silvio L., Jayakumar P. Cellular response to osteoinductive materials in orthopedic surgery. In: Di Silvo L., editor. Cellular Response to Biomaterials. Volume 1. Woodhead Publishing; Cambridge, UK: 2009. pp. 313–343. - DOI
-
- Şelaru A., Drăgușin D.-M., Olăreț E., Serafim A., Steinmüller-Nethl D., Vasile E., Iovu H., Stancu I.-C., Costache M., Dinescu S. Fabrication and Biocompatibility Evaluation of Nanodiamonds-Gelatin Electrospun Materials Designed for Prospective Tissue Regeneration Applications. Materials. 2019;12:2933. doi: 10.3390/ma12182933. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
- Project no. 154/25.11.2016, P_37_221/2015, SMIS code 108117, "A novel graphene biosensor testing osteogenic potency; capturing best stem cell performance for regenerative medicine" (GRABTOP)/National Authority for Scientific Research and Innovation, Operational Program Competitiveness Axis 1-Section E Program co-funded from European Regional Development Fund "Investments for Your Future"
- PN-III-P1-1.2-PCCDI-2017-0782/REGMED/Unitatea Executiva Pentru Finantarea Invatamantului Superior a Cercetarii Dezvoltarii si Inovarii
LinkOut - more resources
Full Text Sources

