Effects of Loigolactobacillus coryniformis K8 CECT 5711 on the Immune Response of Elderly Subjects to COVID-19 Vaccination: A Randomized Controlled Trial
- PMID: 35011103
- PMCID: PMC8747230
- DOI: 10.3390/nu14010228
Effects of Loigolactobacillus coryniformis K8 CECT 5711 on the Immune Response of Elderly Subjects to COVID-19 Vaccination: A Randomized Controlled Trial
Abstract
Elderly people are particularly vulnerable to COVID-19, with a high risk of developing severe disease and a reduced immune response to the COVID-19 vaccine. A randomized, placebo-controlled, double-blind trial to assess the effect of the consumption of the probiotic Loigolactobacillus coryniformis K8 CECT 5711 on the immune response generated by the COVID-19 vaccine in an elderly population was performed. Two hundred nursing home residents >60 yrs that had not COVID-19 were randomized to receive L. coryniformis K8 or a placebo daily for 3 months. All volunteers received a complete vaccination schedule of a mRNA vaccine, starting the intervention ten days after the first dose. Specific IgG and IgA antibody levels were analyzed 56 days after the end of the immunization process. No differences between the groups were observed in the antibody levels. During the intervention, 19 subjects had COVID-19 (11 receiving K8 vs. 8 receiving placebo, p = 0.457). Subgroup analysis in these patients showed that levels of IgG were significantly higher in those receiving K8 compared to placebo (p = 0.038). Among subjects >85 yrs that did not get COVID-19, administration of K8 tended to increase the IgA levels (p = 0.082). The administration of K8 may enhance the specific immune response against COVID-19 and may improve the COVID-19 vaccine-specific responses in elderly populations.
Keywords: COVID-19; elderly; immune response; probiotic.
Conflict of interest statement
J.A.M.-L., C.R., O.B., M.O. and R.B.-R. are workers of Biosearch Life, a Kerry company, owner of the patent of
Figures

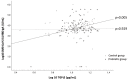
References
-
- World Health Organization; Geneva: 2020. [(accessed on 15 October 2021)]. WHO COVID-19 Dashboard. Available online: https://covid19.who.int/
-
- Docherty A., Harrison E., Green C., Hardwick H., Pius R., Norman L., Holden K., Read J., Dondelinger F., Carson G. Features of 16,749 Hospitalised UK Patients with COVID-19 Using the ISARIC WHO Clinical Characterisation Protocol. [(accessed on 15 October 2021)];2020 Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7243036/ - PMC - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

