Copper(II) Phenanthroline-Based Complexes as Potential AntiCancer Drugs: A Walkthrough on the Mechanisms of Action
- PMID: 35011273
- PMCID: PMC8746828
- DOI: 10.3390/molecules27010049
Copper(II) Phenanthroline-Based Complexes as Potential AntiCancer Drugs: A Walkthrough on the Mechanisms of Action
Abstract
Copper is an endogenous metal ion that has been studied to prepare a new antitumoral agent with less side-effects. Copper is involved as a cofactor in several enzymes, in ROS production, in the promotion of tumor progression, metastasis, and angiogenesis, and has been found at high levels in serum and tissues of several types of human cancers. Under these circumstances, two strategies are commonly followed in the development of novel anticancer Copper-based drugs: the sequestration of free Copper ions and the synthesis of Copper complexes that trigger cell death. The latter strategy has been followed in the last 40 years and many reviews have covered the anticancer properties of a broad spectrum of Copper complexes, showing that the activity of these compounds is often multi factored. In this work, we would like to focus on the anticancer properties of mixed Cu(II) complexes bearing substituted or unsubstituted 1,10-phenanthroline based ligands and different classes of inorganic and organic auxiliary ligands. For each metal complex, information regarding the tested cell lines and the mechanistic studies will be reported and discussed. The exerted action mechanisms were presented according to the auxiliary ligand/s, the metallic centers, and the increasing complexity of the compound structures.
Keywords: 1,10-phenanthroline; anticancer chemotherapy; cancer; cell stress response; chemoresistance; coordination compounds; copper.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
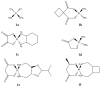
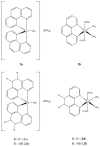
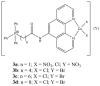

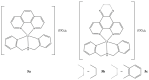



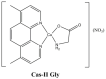
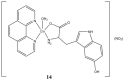

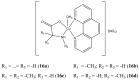
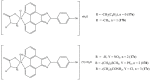



References
-
- Arnesano F., Natile G. Mechanistic insight into the cellular uptake and processing of cisplatin 30 years after its approval by FDA. Coord. Chem. Rev. 2009;253:2070–2081. doi: 10.1016/j.ccr.2009.01.028. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

