Bone Marrow Harbors a Unique Population of Dendritic Cells with the Potential to Boost Neutrophil Formation upon Exposure to Fungal Antigen
- PMID: 35011617
- PMCID: PMC8750392
- DOI: 10.3390/cells11010055
Bone Marrow Harbors a Unique Population of Dendritic Cells with the Potential to Boost Neutrophil Formation upon Exposure to Fungal Antigen
Abstract
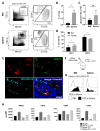
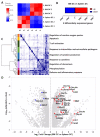
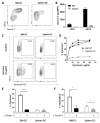
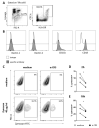
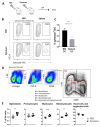
Apart from controlling hematopoiesis, the bone marrow (BM) also serves as a secondary lymphoid organ, as it can induce naïve T cell priming by resident dendritic cells (DC). When analyzing DCs in murine BM, we uncovered that they are localized around sinusoids, can (cross)-present antigens, become activated upon intravenous LPS-injection, and for the most part belong to the cDC2 subtype which is associated with Th2/Th17 immunity. Gene-expression profiling revealed that BM-resident DCs are enriched for several c-type lectins, including Dectin-1, which can bind beta-glucans expressed on fungi and yeast. Indeed, DCs in BM were much more efficient in phagocytosis of both yeast-derived zymosan-particles and Aspergillus conidiae than their splenic counterparts, which was highly dependent on Dectin-1. DCs in human BM could also phagocytose zymosan, which was dependent on β1-integrins. Moreover, zymosan-stimulated BM-resident DCs enhanced the differentiation of hematopoietic stem and progenitor cells towards neutrophils, while also boosting the maintenance of these progenitors. Our findings signify an important role for BM DCs as translators between infection and hematopoiesis, particularly in anti-fungal immunity. The ability of BM-resident DCs to boost neutrophil formation is relevant from a clinical perspective and contributes to our understanding of the increased susceptibility for fungal infections following BM damage.
Keywords: bone marrow; dectin-1; dendritic cells; fungal infection; granulopoiesis; hematopoiesis; zymosan.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Milo I., Sapoznikov A., Kalchenko V., Tal O., Krauthgamer R., Van Rooijen N., Dudziak D., Jung S., Shakhar G. Dynamic imaging reveals promiscuous crosspresentation of blood-borne antigens to naïve CD8+ T cells in the bone marrow. Blood. 2013;122:193–208. doi: 10.1182/blood-2012-01-401265. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

