A Tight Control of Non-Canonical TGF-β Pathways and MicroRNAs Downregulates Nephronectin in Podocytes
- PMID: 35011710
- PMCID: PMC8750045
- DOI: 10.3390/cells11010149
A Tight Control of Non-Canonical TGF-β Pathways and MicroRNAs Downregulates Nephronectin in Podocytes
Abstract
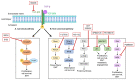
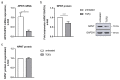
Nephronectin (NPNT) is an extracellular matrix protein in the glomerular basement membrane that is produced by podocytes and is important for the integrity of the glomerular filtration barrier. Upregulated transforming growth factor β (TGF-β) and altered NPNT are seen in different glomerular diseases. TGF-β downregulates NPNT and upregulates NPNT-targeting microRNAs (miRs). However, the pathways involved were previously unknown. By using selective inhibitors of the canonical, SMAD-dependent, and non-canonical TGF-β pathways, we investigated NPNT transcription, translation, secretion, and regulation through miRs in podocytes. TGF-β decreased NPNT mRNA and protein in cultured human podocytes. TGF-β-dependent regulation of NPNT was meditated through intracellular signaling pathways. Under baseline conditions, non-canonical pathways predominantly regulated NPNT post-transcriptionally. Podocyte NPNT secretion, however, was not dependent on canonical or non-canonical TGF-β pathways. The canonical TGF-β pathway was also dispensable for NPNT regulation after TGF-β stimulation, as TGF-β was still able to downregulate NPNT in the presence of SMAD inhibitors. In contrast, in the presence of different non-canonical pathway inhibitors, TGF-β stimulation did not further decrease NPNT expression. Moreover, distinct non-canonical TGF-β pathways mediated TGF-β-induced upregulation of NPNT-targeting miR-378a-3p. Thus, we conclude that post-transcriptional fine-tuning of NPNT expression in podocytes is mediated predominantly through non-canonical TGF-β pathways.
Keywords: TGF-β; microRNAs; nephronectin; podocytes; podocytopathies; post-transcriptional regulation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
Podocytes regulate the glomerular basement membrane protein nephronectin by means of miR-378a-3p in glomerular diseases.Kidney Int. 2017 Oct;92(4):836-849. doi: 10.1016/j.kint.2017.03.005. Epub 2017 May 3. Kidney Int. 2017. PMID: 28476557 Free PMC article.
-
Glomerular Endothelial Cell-Derived microRNA-192 Regulates Nephronectin Expression in Idiopathic Membranous Glomerulonephritis.J Am Soc Nephrol. 2021 Nov;32(11):2777-2794. doi: 10.1681/ASN.2020121699. J Am Soc Nephrol. 2021. PMID: 34716242 Free PMC article.
-
Foreseeing the future of glomerular disease through slits: miR-NPNT axis.Kidney Int. 2017 Oct;92(4):782-784. doi: 10.1016/j.kint.2017.04.036. Kidney Int. 2017. PMID: 28938947
-
Mechanisms and consequences of TGF-ß overexpression by podocytes in progressive podocyte disease.Cell Tissue Res. 2012 Jan;347(1):129-40. doi: 10.1007/s00441-011-1169-7. Epub 2011 May 4. Cell Tissue Res. 2012. PMID: 21541658 Free PMC article. Review.
-
Effects of TGF-beta on podocyte growth and disease progression in proliferative podocytopathies.Kidney Blood Press Res. 2010;33(1):24-9. doi: 10.1159/000285844. Epub 2010 Feb 23. Kidney Blood Press Res. 2010. PMID: 20185928 Review.
Cited by
-
Urolithin A Ameliorates the TGF Beta-Dependent Impairment of Podocytes Exposed to High Glucose.J Pers Med. 2024 Aug 28;14(9):914. doi: 10.3390/jpm14090914. J Pers Med. 2024. PMID: 39338168 Free PMC article.
-
Characterizing Intraindividual Podocyte Morphology In Vitro with Different Innovative Microscopic and Spectroscopic Techniques.Cells. 2023 Apr 25;12(9):1245. doi: 10.3390/cells12091245. Cells. 2023. PMID: 37174644 Free PMC article.
-
The context-dependent role of transforming growth factor-β/miR-378a-3p/connective tissue growth factor in vascular calcification: a translational study.Aging (Albany NY). 2023 Feb 13;15(3):830-845. doi: 10.18632/aging.204518. Epub 2023 Feb 13. Aging (Albany NY). 2023. PMID: 36787443 Free PMC article.
References
-
- Byron A., Randles M.J., Humphries J.D., Mironov A., Hamidi H., Harris S., Mathieson P.W., Saleem M.A., Satchell S.C., Zent R., et al. Glomerular cell cross-talk influences composition and assembly of extracellular matrix. J. Am. Soc. Nephrol. 2014;25:953–966. doi: 10.1681/ASN.2013070795. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

