[Tumor necrosis factor-α promotes osteoclast differentiation via sialylation in mice]
- PMID: 35012907
- PMCID: PMC8752434
- DOI: 10.12122/j.issn.1673-4254.2021.12.03
[Tumor necrosis factor-α promotes osteoclast differentiation via sialylation in mice]
Abstract
Objective: To explore the mechanism through which tumor necrosis factor-α (TNF-α) promotes osteoclast differentiation.
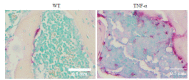
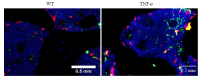
Methods: Bilateral knee joint samples were collected from 4-month-old wild-type mice and TNF-α transgenic mice for CT scan analysis, TRAP staining and sialic acid staining analysis. The osteoclast precursor (RAW264.7) cells were cultured for 3 days in induction medium in the presence of vehicle, TNF-α, or TNF-α and sialidase, and were then examined with RT-qPCR, TRAP staining, and sialic acid immunofluorescence co-localization staining. Bone marrow-derived macrophages isolated from the wild-type mice and TNF-α transgenic mouse and cultured in induction medium with or without the addition of sialidase, and TRAP and sialic acid staining was performed after 3 days of cell culture.
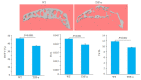
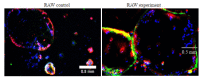
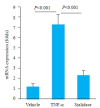
Results: TRAP staining showed that the number of osteoclasts increased significantly in TNF-α transgenic mice as compared with the wild-type mice (P < 0.0001), and micro-CT analysis revealed significant reductions of BV/TV, Tb.N, and Tb.Th in TNF-α transgenic mice (P < 0.001). The osteoclasts in TNF-α transgenic mice also showed a significantly increased expression of sialic acid (P=0.004). In the cell experiment, RAW264.7 cells cultured with TNF-α showed a significantly higher expression of sialic acid (P < 0.0001) and a greater osteoclast formation rate (P=0.0007) than the the control cells, while the addition of sialidase significantly reduced sialic acid expression, osteoclast formation rate and TRAP mRNA level in TNF-α-treated cells (P < 0.0001). Similarly, in the bone marrow-derived macrophages, sialic acid expression and osteoclast formation rate were significantly increased by incubation with TNF-α (P < 0.0001), but the increments were obviously reduced by addition of sialidase in the medium (P < 0.0001).
Conclusion: TNF-α can promote the differentiation and activity of osteoclasts by increasing the sialylation level in the osteoclasts.
目的: 探究TNF-α促进破骨细胞分化的作用机制。
方法: 利用4月龄野生型小鼠与同龄TNF-α转基因鼠进行体内研究,分别收集双侧膝骨关节样本后,进行CT扫描分析骨量差异,TRAP染色及免疫荧光染色分析破骨细胞分化程度及唾液酸表达水平。体外培养破骨细胞前体细胞系RAW264.7分为3组:RAW对照组(不含药物的分化培养基处理);RAW实验组(含有TNF-α的分化培养基处理);RAW药物组(含有TNF-α和唾液酸酶的分化培养基处理)。体外培养3 d后进行qPCR检测,TRAP染色,唾液酸免疫荧光共定位染色分析。同时,分离培养野生型小鼠(BM对照组)和TNF-α转基因鼠[BM实验组(不含药物的分化培养基处理),BM药物组(含有唾液酸酶的分化培养基处理)]原代骨髓源巨噬细胞,3 d后进行TRAP染色及唾液酸染色。
结果: TRAP染色显示,TNF-α转基因鼠膝关节软骨下骨中破骨细胞数量较野生型小鼠显著增多(P < 0.001);其骨量/体积(BV/TV)、骨小梁数量(Tb.N)、骨小梁厚度(Tb.Th)则显著降低(P < 0.001);而TNF-α转基因鼠软骨下骨中唾液酸与TRAP荧光共定位阳性细胞数也显著高于野生型小鼠(P=0.004)。RAW实验组唾液酸表达平均荧光强度(P < 0.001)及破骨细胞形成率(P < 0.001)均显著高于RAW对照组,而RAW药物组唾液酸表达平均荧光强度、破骨细胞形成率以及TRAP基因转录水平显著低于RAW实验组(P < 0.001)。BM实验组较BM对照组唾液酸表达平均荧光强度、破骨细胞形成率均显著提高(P < 0.001),而BM药物组较BM实验组唾液酸表达平均荧光强度、破骨细胞形成率均显著降低(P < 0.001)。
结论: TNF-α可通过提高破骨细胞的唾液酸化水平进而促进破骨细胞的分化和功能。
Keywords: osteoclasts; sialylation; tumor necrosis factor-α.
Figures




References
-
- Locksley RM, Killeen N, Lenardo MJ. The TNF and TNF receptor superfamilies: integrating mammalian biology. Cell. 2001;104(4):487–501. doi: 10.1016/S0092-8674(01)00237-9. [Locksley RM, Killeen N, Lenardo MJ. The TNF and TNF receptor superfamilies: integrating mammalian biology[J]. Cell, 2001, 104 (4): 487-501.] - DOI - PubMed
-
- Ibáñez L, Abou EG, Ciucci T, et al. Inflammatory Osteoclasts Prime TNFα-Producing CD4+ T Cells and Express CX3 CR1. J Bone Miner Res. 2016;31(10):1899–908. doi: 10.1002/jbmr.2868. [Ibáñez L, Abou EG, Ciucci T, et al. Inflammatory Osteoclasts Prime TNFα-Producing CD4+ T Cells and Express CX3 CR1[J]. J Bone Miner Res, 2016, 31(10): 1899-908.] - DOI - PubMed
-
- Lee SE, Chung WJ, Kwak HB, et al. Tumor necrosis factor-alpha supports the survival of osteoclasts through the activation of Akt and ERK. J Biol Chem. 2001;276(52):49343–9. doi: 10.1074/jbc.M103642200. [Lee SE, Chung WJ, Kwak HB, et al. Tumor necrosis factor-alpha supports the survival of osteoclasts through the activation of Akt and ERK[J]. J Biol Chem, 2001, 276(52): 49343-9.] - DOI - PubMed
-
- Dougall WC, Glaccum M, Charrier K, et al. RANK is essential for osteoclast and lymph node development. Genes Dev. 1999;13(18):2412–24. doi: 10.1101/gad.13.18.2412. [Dougall WC, Glaccum M, Charrier K, et al. RANK is essential for osteoclast and lymph node development[J]. Genes Dev, 1999, 13 (18): 2412-24.] - DOI - PMC - PubMed
-
- Kim D, Mebius RE, MacMicking JD, et al. Regulation of peripheral lymph node Genesis by the tumor necrosis factor family member TRANCE. J Exp Med. 2000;192(10):1467–78. doi: 10.1084/jem.192.10.1467. [Kim D, Mebius RE, MacMicking JD, et al. Regulation of peripheral lymph node Genesis by the tumor necrosis factor family member TRANCE[J]. J Exp Med, 2000, 192(10): 1467-78.] - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
