Systematic analysis of drug-associated myocarditis reported in the World Health Organization pharmacovigilance database
- PMID: 35013204
- PMCID: PMC8748719
- DOI: 10.1038/s41467-021-27631-8
Systematic analysis of drug-associated myocarditis reported in the World Health Organization pharmacovigilance database
Abstract
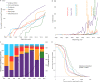
While multiple pharmacological drugs have been associated with myocarditis, temporal trends and overall mortality have not been reported. Here we report the spectrum and main features of 5108 reports of drug-induced myocarditis, in a worldwide pharmacovigilance analysis, comprising more than 21 million individual-case-safety reports from 1967 to 2020. Significant association between myocarditis and a suspected drug is assessed using disproportionality analyses, which use Bayesian information component estimates. Overall, we identify 62 drugs associated with myocarditis, 41 of which are categorized into 5 main pharmacological classes: antipsychotics (n = 3108 reports), salicylates (n = 340), antineoplastic-cytotoxics (n = 190), antineoplastic-immunotherapies (n = 538), and vaccines (n = 790). Thirty-eight (61.3%) drugs were not previously reported associated with myocarditis. Antipsychotic was the first (1979) and most reported class (n = 3018). In 2019, the two most reported classes were antipsychotics (54.7%) and immunotherapies (29.5%). Time-to-onset between treatment start and myocarditis is 15 [interquartile range: 10; 23] days. Subsequent mortality is 10.3% and differs between drug classes with immunotherapies the highest, 32.5% and salicylates the lowest, 2.6%. These elements highlight the diversity of presentations of myocarditis depending on drug class, and show the emerging role of antineoplastic drugs in the field of drug-induced myocarditis.
© 2022. The Author(s).
Conflict of interest statement
M.K. has received consulting fees from Sanofi, lecture fees from Bayer, a research grant from PHRC 2015 (P150921), Federation Française de Cardiologie. J.S. reports during the past 2 years the following disclosures: consulting Fees or Lecture Fees or Travel Support from AstraZeneca, Bayer HealthCare SAS, Boehringer Ingelheim France, CSL Behring SA, Gilead Science, Sanofi-Aventis France, Terumo France SAS, Abbott Medical France SAS, Stockholder of Pharmaseeds. B.B.M. has no conflict of interest regarding this work, and is currently employed by Kayrros (Paris, France). E. G. received consulting fees from consulting fees from Boston Scientific, Microport and Medtronic. J.J.M. received fees from Pfizer, Novartis, Bristol-Myers Squibb, Deciphera, Audentes Pharmaceuticals, Nektar, Takeda, Ipsen, Myokardia, AstraZeneca, GlaxoSmithKline, Intrexon, and Regeneron, and is supported by R01 HL141466. J.-E. S. has participated to BMS advisory boards. The remaining authors declare no competing interests.
Figures



References
-
- Vos T, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2015;386:743–800. doi: 10.1016/S0140-6736(15)60692-4. - DOI - PMC - PubMed
-
- James SL, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392:1789–1858. doi: 10.1016/S0140-6736(18)32279-7. - DOI - PMC - PubMed

