Surface-Directed Mineralization of Fibrous Collagen Scaffolds in Simulated Body Fluid for Tissue Engineering Applications
- PMID: 35014369
- PMCID: PMC8978552
- DOI: 10.1021/acsabm.0c01507
Surface-Directed Mineralization of Fibrous Collagen Scaffolds in Simulated Body Fluid for Tissue Engineering Applications
Abstract
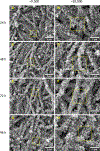
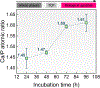
The use of polymer additives that stabilize fluidic amorphous calcium phosphate is key to obtaining intrafibrillar mineralization of collagen in vitro. On the other hand, this biomimetic approach inhibits the nucleation of mineral crystals in unconfined extrafibrillar spaces, that is, extrafibrillar mineralization. The extrafibrillar mineral content is a significant feature to replicate from hard connective tissues such as bone and dentin as it contributes to the final microarchitecture and mechanical stiffness of the biomineral composite. Herein, we report a straightforward route to produce densely mineralized collagenous composites via a surface-directed process devoid of the aid of polymer additives. Simulated body fluid (1×) is employed as a biomimetic crystallizing medium, following a preloading procedure on the collagen surface to quickly generate the amorphous precursor species required to initiate matrix mineralization. This approach consistently leads to the formation of extrafibrillar bioactive minerals in bulk collagen scaffolds, which may offer an advantage in the production of osteoconductive collagen-apatite materials for tissue engineering and repair purposes.
Keywords: amorphous precursor; apatite; mineralization; nanofibrous scaffolds; type-I collagen.
Figures




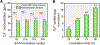


Similar articles
-
Synergistic intrafibrillar/extrafibrillar mineralization of collagen scaffolds based on a biomimetic strategy to promote the regeneration of bone defects.Int J Nanomedicine. 2016 May 12;11:2053-67. doi: 10.2147/IJN.S102844. eCollection 2016. Int J Nanomedicine. 2016. PMID: 27274235 Free PMC article.
-
Fabrication of intrafibrillar and extrafibrillar mineralized collagen/apatite scaffolds with a hierarchical structure.J Biomed Mater Res A. 2016 May;104(5):1153-61. doi: 10.1002/jbm.a.35649. Epub 2016 Feb 2. J Biomed Mater Res A. 2016. PMID: 26748775
-
Biomimetic collagen-hydroxyapatite composite fabricated via a novel perfusion-flow mineralization technique.Tissue Eng Part C Methods. 2013 Jul;19(7):487-96. doi: 10.1089/ten.TEC.2012.0452. Epub 2013 Jan 4. Tissue Eng Part C Methods. 2013. PMID: 23157544 Free PMC article.
-
Biomineralization of Collagen-Based Materials for Hard Tissue Repair.Int J Mol Sci. 2021 Jan 19;22(2):944. doi: 10.3390/ijms22020944. Int J Mol Sci. 2021. PMID: 33477897 Free PMC article. Review.
-
In Vitro Mineralization of Collagen.Adv Mater. 2021 Apr;33(16):e2004418. doi: 10.1002/adma.202004418. Epub 2021 Mar 12. Adv Mater. 2021. PMID: 33711177 Free PMC article. Review.
Cited by
-
Regulatory potency of oligomeric proanthocyanidins (PACs) on extrafibrillar and intrafibrillar collagen mineralization.J Appl Polym Sci. 2025 Apr 10;142(14):e56694. doi: 10.1002/app.56694. Epub 2025 Jan 22. J Appl Polym Sci. 2025. PMID: 40799984
-
Preparation and characterization of FsHA/FsCol beads: Cell attachment and cytotoxicity studies.Heliyon. 2023 May 1;9(5):e15838. doi: 10.1016/j.heliyon.2023.e15838. eCollection 2023 May. Heliyon. 2023. PMID: 37206015 Free PMC article.
References
-
- Weiner S; Wagner HD THE MATERIAL BONE: Structure-Mechanical Function Relations. Annu. Rev. Mater. Sci 1998, 28 (1), 271–298. 10.1146/annurev.matsci.28.1.271. - DOI
-
- Leonor IB; Rodrigues AI; Reis RL Designing Biomaterials Based on Biomineralization for Bone Repair and Regeneration. In Biomineralization and Biomaterials; Elsevier, 2016; pp 377–404. 10.1016/B978-1-78242-338-6.00014-4. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
