Golden Syrian hamster as a model to study cardiovascular complications associated with SARS-CoV-2 infection
- PMID: 35014610
- PMCID: PMC8794466
- DOI: 10.7554/eLife.73522
Golden Syrian hamster as a model to study cardiovascular complications associated with SARS-CoV-2 infection
Abstract
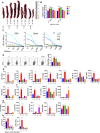
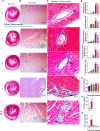
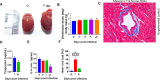
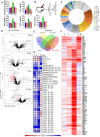
Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection in the Golden Syrian hamster causes lung pathology that resembles human coronavirus disease (COVID-19). However, extrapulmonary pathologies associated with SARS-CoV-2 infection and post-COVID sequelae remain to be understood. Here, we show, using a hamster model, that the early phase of SARS-CoV-2 infection leads to an acute inflammatory response and lung pathologies, while the late phase of infection causes cardiovascular complications (CVCs) characterized by ventricular wall thickening associated with increased ventricular mass/body mass ratio and interstitial coronary fibrosis. Molecular profiling further substantiated our findings of CVC as SARS-CoV-2-infected hamsters showed elevated levels of serum cardiac troponin I, cholesterol, low-density lipoprotein, and long-chain fatty acid triglycerides. Serum metabolomics profiling of SARS-CoV-2-infected hamsters identified N-acetylneuraminate, a functional metabolite found to be associated with CVC, as a metabolic marker was found to be common between SARS-CoV-2-infected hamsters and COVID-19 patients. Together, we propose hamsters as a suitable animal model to study post-COVID sequelae associated with CVC, which could be extended to therapeutic interventions.
Keywords: SARS-CoV2; cardiovascular; hamster; infectious disease; microbiology.
© 2022, Rizvi et al.
Conflict of interest statement
ZR, RD, SS, AB, JD, YK, TS, SG, SA, MT, DR, AY, GM, AP, SS, SA, AA No competing interests declared
Figures





References
-
- Anantharaj A, Gujjar S, Kumar S, Verma N, Wangchuk J, Khan NA, Panwar A, Kanakan AAV, Vasudevan JS, Das A, Pandey AK, Pandey R, Medigeshi GR. Kinetics of Viral Load, Immunological Mediators and Characterization of a SARS-CoV-2 Isolate in Mild COVID-19 Patients during Acute Phase of Infection. medRxiv. 2020 doi: 10.1101/2020.11.05.20226621. - DOI
-
- Boudewijns R, Thibaut HJ, Kaptein SJF, Li R, Vergote V, Seldeslachts L, Van Weyenbergh J, De Keyzer C, Bervoets L, Sharma S, Liesenborghs L, Ma J, Jansen S, Van Looveren D, Vercruysse T, Wang X, Jochmans D, Martens E, Roose K, De Vlieger D, Schepens B, Van Buyten T, Jacobs S, Liu Y, Martí-Carreras J, Vanmechelen B, Wawina-Bokalanga T, Delang L, Rocha-Pereira J, Coelmont L, Chiu W, Leyssen P, Heylen E, Schols D, Wang L, Close L, Matthijnssens J, Van Ranst M, Compernolle V, Schramm G, Van Laere K, Saelens X, Callewaert N, Opdenakker G, Maes P, Weynand B, Cawthorne C, Vande Velde G, Wang Z, Neyts J, Dallmeier K. STAT2 signaling restricts viral dissemination but drives severe pneumonia in SARS-CoV-2 infected hamsters. Nature Communications. 2020;11:5838. doi: 10.1038/s41467-020-19684-y. - DOI - PMC - PubMed
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

