Photosystem II antenna modules CP43 and CP47 do not form a stable 'no reaction centre complex' in the cyanobacterium Synechocystis sp. PCC 6803
- PMID: 35015206
- PMCID: PMC9458580
- DOI: 10.1007/s11120-022-00896-w
Photosystem II antenna modules CP43 and CP47 do not form a stable 'no reaction centre complex' in the cyanobacterium Synechocystis sp. PCC 6803
Abstract
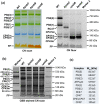
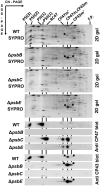
The repair of photosystem II is a key mechanism that keeps the light reactions of oxygenic photosynthesis functional. During this process, the PSII central subunit D1 is replaced with a newly synthesized copy while the neighbouring CP43 antenna with adjacent small subunits (CP43 module) is transiently detached. When the D2 protein is also damaged, it is degraded together with D1 leaving both the CP43 module and the second PSII antenna module CP47 unassembled. In the cyanobacterium Synechocystis sp. PCC 6803, the released CP43 and CP47 modules have been recently suggested to form a so-called no reaction centre complex (NRC). However, the data supporting the presence of NRC can also be interpreted as a co-migration of CP43 and CP47 modules during electrophoresis and ultracentrifugation without forming a mutual complex. To address the existence of NRC, we analysed Synechocystis PSII mutants accumulating one or both unassembled antenna modules as well as Synechocystis wild-type cells stressed with high light. The obtained results were not compatible with the existence of a stable NRC since each unassembled module was present as a separate protein complex with a mutually similar electrophoretic mobility regardless of the presence of the second module. The non-existence of NRC was further supported by isolation of the His-tagged CP43 and CP47 modules from strains lacking either D1 or D2 and their migration patterns on native gels.
Keywords: CP43; CP47; No reaction centre complex; Photosynthesis; Photosystem II.
© 2022. The Author(s).
Conflict of interest statement
The authors have no conflict of interest to declare that are relevant to the content of this article.
Figures


References
-
- Barber J. Molecular Basis of the Vulnerability of Photosystem II to Damage by Light. Austr J Plant Physiol. 1995;22:201–208. doi: 10.1071/PP9950201. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

