Evaluation of the frailty characteristics and clinical outcomes according to the new frailty-based outcome prediction model (Myeloma Risk Profile-MRP) in a UK real-world cohort of elderly newly diagnosed Myeloma patients
- PMID: 35015781
- PMCID: PMC8752006
- DOI: 10.1371/journal.pone.0262388
Evaluation of the frailty characteristics and clinical outcomes according to the new frailty-based outcome prediction model (Myeloma Risk Profile-MRP) in a UK real-world cohort of elderly newly diagnosed Myeloma patients
Abstract
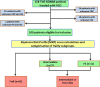
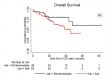
The management of myeloma in the elderly is shifting its focus towards reducing the risk of under-treating fit patients and the risk of over-treating frail patients. Frailty assessment is required in this patient group in order to individualise treatment decisions. In addition to the proven prognostic values of the International Myeloma Working Group (IMWG) frailty score and the revised Myeloma Co-morbidity Index (R-MCI), a new easy-to-use frailty-based risk profile score (high-risk (i.e. frail), medium risk (i.e. intermediate-fitness) and low-risk (i.e. fit)) named Myeloma Risk Profile (MRP) was shown to be predictive of survival in the clinical trial setting. In this retrospective real-world study, we set out to evaluate the frailty characteristics and clinical outcomes according to the different MRP scoring algorithm categories (frail vs. intermediate vs fit), in a high risk cohort of elderly newly diagnosed myeloma patients treated with the fixed-duration triplet therapy VCD (bortezomib with cyclophosphamide and dexamethasone). Clinical outcomes included: reason for treatment discontinuation, overall response rate (ORR), overall survival (OS), progression-free survival (PFS), and adverse events (AEs). Out of 100 patients, 62 were frail, 27 were intermediate and 11 were fit, according to MRP scores. To enable meaningful comparisons between comparable numbers, subgroups analyses for ORR, OS, PFS, and AEs focused on frail (n = 62) versus intermediate or fit (n = 38) patients. The proportion of patients in each subgroup who were able to complete the planned course of treatment was (frail: 43.5% vs. intermediate or fit: 55.3%). A higher proportion in the frail subgroup discontinued therapy due to progressive disease (19.4% vs. 2.6%). Discontinuation due to toxicity was comparable across subgroups (14.5% vs. 15.8%), ORR in the total cohort was 75%, and this was comparable between subgroups (frail: 74.2% vs. intermediate or fit: 76.3%). There was a trend for a shorter median OS in the frail subgroup but without a statistical significance: (frail vs. intermediate or fit): (46 months vs. not reached, HR: 1.94, 95% CI 0.89-4.2, p = 0.094). There was no difference in median PFS between subgroups: (frail vs. intermediate or fit): (11.8 vs. 9.9 months, HR: 0.99, 95% CI: 0.61-1.61, P = 0.982). This cohort demonstrated a higher incidence rate of AEs in frail patients compared to those in the intermediate or fit group: patients with at least one any grade toxicity (85.5% vs. 71.1%), patients with at least one ≥G3 AE (37.1% vs. 21.1%). In conclusion, our study is to the first to evaluate clinical outcomes according to MRP in a high risk real-world cohort of patients treated exclusively with the proteasome inhibitor-based VCD therapy. Our study demonstrated a trend for worse OS in addition to worse AE outcomes in the frail group, but no difference in PFS with this fixed-duration therapy. MRP is an easy-to-use tool in clinical practice; its prognostic value was validated in the real-world in a large cohort of patients from the Danish Registry. Further evaluation of MRP in the real-world when continuous therapies are used, can further support the generalisability of its prognostic value in elderly myeloma patients.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

References
-
- Cancer Research UK. Cancer statistics: Myeloma incidence statistics. Available from: http://www.cancerresearchuk.org/health-professional/cancer-statistics/st.... Accessed January 21st, 2021.
-
- Engelhardt M, Ihorst G, Duque-Afonso J, Wedding U, Spät-Schwalbe E, Goede V, et al.. Structured assessment of frailty in multiple myeloma as a paradigm of individualized treatment algorithms in cancer patients at advanced age. Haematologica. 2020. May; 105(5): 1183–1188. doi: 10.3324/haematol.2019.242958 - DOI - PMC - PubMed
-
- Engelhardt M, Dold SM, Ihorst G, et al.. Geriatric assessment in multiple myeloma patients: validation of the International Myeloma Working Group (IMWG) score and comparison with other common comorbidity scores. Haematologica 2016; 101: 1110–19. doi: 10.3324/haematol.2016.148189 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

