Proprioceptive sensitivity to imposed finger deflections
- PMID: 35020504
- PMCID: PMC8799383
- DOI: 10.1152/jn.00513.2021
Proprioceptive sensitivity to imposed finger deflections
Abstract
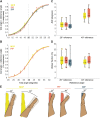
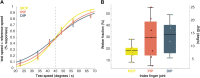
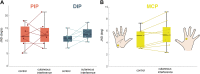
Hand proprioception, the sense of the posture and movements of the wrist and digits, is critical to dexterous manual behavior and to stereognosis, the ability to sense the three-dimensional structure of objects held in the hand. To better understand this sensory modality and its role in hand function, we sought to characterize the acuity with which the postures and movements of finger joints are sensed. To this end, we measured the ability of human subjects to discriminate changes in posture and speed around the three joints of the index finger. In these experiments, we isolated the sensory component by imposing the postures on an otherwise still hand, to complement other studies in which subjects made judgments on actively achieved postures. We found that subjects could reliably sense 12-16% changes in joint angle and 18-32% changes in joint speed. Furthermore, the acuity for posture and speed was comparable across the three joints of the finger. Finally, task performance was unaffected by the presence of a vibratory stimulus, calling into question the role of cutaneous cues in hand proprioception.NEW & NOTEWORTHY Manual dexterity and stereognosis are supported by two exquisite sensory systems, namely touch and proprioception. Here, we measure the sensitivity of hand proprioception and show that humans can sense the posture and movements of the fingers with great accuracy. We also show that application of a skin vibration does not impair sensitivity, suggesting that proprioceptive acuity relies primarily on receptors in the muscles (and possibly tendons) rather than the skin.
Keywords: angular acuity; angular speed; muscle spindles; psychophysics.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures


Similar articles
-
Movement detection at the distal joint of the human thumb and fingers.Exp Brain Res. 1998 Sep;122(1):85-92. doi: 10.1007/s002210050494. Exp Brain Res. 1998. PMID: 9772115
-
Finger Posture and Finger Load are Perceived Independently.Sci Rep. 2019 Oct 21;9(1):15031. doi: 10.1038/s41598-019-51131-x. Sci Rep. 2019. PMID: 31636297 Free PMC article.
-
Representation of wrist joint kinematics by the ensemble of muscle spindles from synergistic muscles.J Neurophysiol. 1998 May;79(5):2265-76. doi: 10.1152/jn.1998.79.5.2265. J Neurophysiol. 1998. PMID: 9582203 Clinical Trial.
-
The proprioceptive senses: their roles in signaling body shape, body position and movement, and muscle force.Physiol Rev. 2012 Oct;92(4):1651-97. doi: 10.1152/physrev.00048.2011. Physiol Rev. 2012. PMID: 23073629 Review.
-
Head and neck position sense.Sports Med. 2008;38(2):101-17. doi: 10.2165/00007256-200838020-00002. Sports Med. 2008. PMID: 18201114 Review.
References
-
- Goodman JM, Bensmaia SJ. The neural basis of haptic perception. In Stevens’ Handbook of Experimental Psychology and Cognitive Neuroscience. Hoboken, NJ: Wiley, 2018, vol. 2, p. 1–39.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

