Arterial intracranial thrombosis as the first manifestation of vaccine-induced immune thrombotic thrombocytopenia (VITT): a case report
- PMID: 35022931
- PMCID: PMC8754523
- DOI: 10.1007/s10072-021-05800-3
Arterial intracranial thrombosis as the first manifestation of vaccine-induced immune thrombotic thrombocytopenia (VITT): a case report
Abstract
Objective: We describe a severe case of vaccine-induced immune thrombotic thrombocytopenia (VITT) after the first dose of the ChAdOx1 nCoV-19 vaccine leading to massive ischemic stroke.
Methods: A 42-year-old woman developed acute left hemiparesis (NIHSS 12) 9 days after the first vaccine dose.
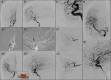
Results: The blood tests revealed low platelets (70 103/μL) and severe increment of D-dimer (70,745 ng/mL FEU). Brain non-contrast computed tomography and multiphasic CT angiography demonstrated a right middle cerebral artery occlusion. The patient was treated with primary thrombectomy, steroids, immunoglobulin, and fondaparinux. Despite the treatment, the neurological status deteriorated and underwent decompressive hemicraniectomy. She was transferred to the rehab's unit 52 days after the onset.
Discussion: Healthcare providers should be aware of the possibility of ischemic stroke as a manifestation of VITT. Awareness on this very rare and possibly fatal complication should be reinforced on both the vaccine recipients and general practitioners.
Keywords: COVID-19; Stroke; Thrombocytopenia; VITT; Vaccine.
© 2021. Fondazione Società Italiana di Neurologia.
Conflict of interest statement
The authors declare no competing interests.
Figures

References
-
- Gresele P, Marietta M, Ageno W, et al. Management of cerebral and splanchnic vein thrombosis associated with thrombocytopenia in subjects previously vaccinated with Vaxzevria (AstraZeneca): a position statement from the Italian Society for the Study of Haemostasis and Thrombosis (SISET) Blood Transfus. 2021 doi: 10.2450/2021.0117-21. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

