Synapsin III gene silencing redeems alpha-synuclein transgenic mice from Parkinson's disease-like phenotype
- PMID: 35038583
- PMCID: PMC9077321
- DOI: 10.1016/j.ymthe.2022.01.021
Synapsin III gene silencing redeems alpha-synuclein transgenic mice from Parkinson's disease-like phenotype
Abstract
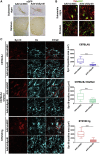
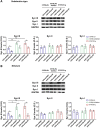
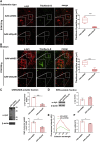
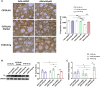
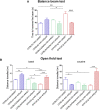
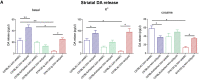
Fibrillary aggregated α-synuclein (α-syn) deposition in Lewy bodies (LB) characterizes Parkinson's disease (PD) and is believed to trigger dopaminergic synaptic failure and a retrograde terminal-to-cell body neuronal degeneration. We described that the neuronal phosphoprotein synapsin III (Syn III) cooperates with α-syn to regulate dopamine (DA) release and can be found in the insoluble α-syn fibrils composing LB. Moreover, we showed that α-syn aggregates deposition, and the associated onset of synaptic deficits and neuronal degeneration occurring following adeno-associated viral vectors-mediated overexpression of human α-syn in the nigrostriatal system are hindered in Syn III knock out mice. This supports that Syn III facilitates α-syn aggregation. Here, in an interventional experimental design, we found that by inducing the gene silencing of Syn III in human α-syn transgenic mice at PD-like stage with advanced α-syn aggregation and overt striatal synaptic failure, we could lower α-syn aggregates and striatal fibers loss. In parallel, we observed recovery from synaptic vesicles clumping, DA release failure, and motor functions impairment. This supports that Syn III consolidates α-syn aggregates, while its downregulation enables their reduction and redeems the PD-like phenotype. Strategies targeting Syn III could thus constitute a therapeutic option for PD.
Keywords: Parkinson's disease; alpha-synuclein; dopamine release; dopaminergic neurons; motor functions; synapsin III; synaptic vesicles.
Copyright © 2022 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors have declared that no conflict of interest exists.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

