Imaging Pulmonary Blood Vessels and Ventilation-Perfusion Mismatch in COVID-19
- PMID: 35041149
- PMCID: PMC8764889
- DOI: 10.1007/s11307-021-01700-2
Imaging Pulmonary Blood Vessels and Ventilation-Perfusion Mismatch in COVID-19
Abstract
COVID-19 hypoxemic patients although sharing a same etiology (SARS-CoV-2 infection) present themselves quite differently from one another. Patients also respond differently to prescribed medicine and to prone Vs supine bed positions. A severe pulmonary ventilation-perfusion mismatch usually triggers moderate to severe COVID-19 cases. Imaging can aid the physician in assessing severity of COVID-19. Although useful for their portability X-ray and ultrasound serving on the frontline to evaluate lung parenchymal abnormalities are unable to provide information about pulmonary vasculature and blood flow redistribution which is a consequence of hypoxemia in COVID-19. Advanced imaging modalities such as computed tomography, single-photon emission tomography, and electrical impedance tomography use a sharp algorithm visualizing pulmonary ventilation-perfusion mismatch in the abnormal and in the apparently normal parenchyma. Imaging helps to access the severity of infection, lung performance, ventilation-perfusion mismatch, and informs strategies for medical treatment. This review summarizes the capacity of these imaging modalities to assess ventilation-perfusion mismatch in COVID-19. Despite having limitations, these modalities provide vital information on blood volume distribution, pulmonary embolism, pulmonary vasculature and are useful to assess severity of lung disease and effectiveness of treatment in COVID-19 patients.
Keywords: COVID-19; CT angiography; Electrical impedance tomography; HRCT; SPECT-CT.
© 2022. World Molecular Imaging Society.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures


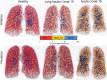



References
-
- Hemmings HC, Egan TD (2019) Pharmacology and physiology for anesthesia: foundations and clinical application. Second edition. ed., Philadelphia, PA: Elsevier, Inc.
-
- Lumb AB, Horner D (2013) Chapter 25—Pulmonary physiology. In Pharmacology and Physiology for anesthesia, Eds. Hemmings HC, Egan TD. Philadelphia: W.B. Saunders, pp 445–457.
-
- Levitzky MG (2018) Pulmonary physiology. Ninth edition. ed., New York: McGraw-Hill Education.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

