The Rapid Antigen Detection Test for SARS-CoV-2 Underestimates the Identification of COVID-19 Positive Cases and Compromises the Diagnosis of the SARS-CoV-2 (K417N/T, E484K, and N501Y) Variants
- PMID: 35047474
- PMCID: PMC8761676
- DOI: 10.3389/fpubh.2021.780801
The Rapid Antigen Detection Test for SARS-CoV-2 Underestimates the Identification of COVID-19 Positive Cases and Compromises the Diagnosis of the SARS-CoV-2 (K417N/T, E484K, and N501Y) Variants
Abstract
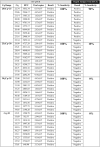
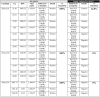
Timely detection of severe acute respiratory syndrome due to coronavirus 2 (SARS-CoV-2) by reverse transcription quantitative polymerase chain reaction (RT-qPCR) has been the gold- strategy for identifying positive cases during the current pandemic. However, faster and less expensive methodologies are also applied for the massive diagnosis of COVID-19. In this way, the rapid antigen test (RAT) is widely used. However, it is necessary to evaluate its detection efficiency considering the current pandemic context with the circulation of new viral variants. In this study, we evaluated the sensitivity and specificity of RAT (SD BIOSENSOR, South Korea), widely used for testing and SARS-CoV-2 diagnosis in Santiago of Chile. The RAT showed a 90% (amplification range of 20 ≤ Cq <25) and 10% (amplification range of 25 ≤ Cq <30) of positive SARS-CoV-2 cases identified previously by RT-qPCR. Importantly, a 0% detection was obtained for samples within a Cq value>30. In SARS-CoV-2 variant detection, RAT had a 42.8% detection sensitivity in samples with RT-qPCR amplification range 20 ≤ Cq <25 containing the single nucleotide polymorphisms (SNP) K417N/T, N501Y and E484K, associated with beta or gamma SARS-CoV-2 variants. This study alerts for the special attention that must be paid for the use of RAT at a massive diagnosis level, especially in the current scenario of appearance of several new SARS-CoV-2 variants which could generate false negatives and the compromise of possible viral outbreaks.
Keywords: COVID-19 diagnosis; COVID-19 false negative rapid test for COVID-19 diagnosis; SARS-CoV-2 detection; pandemic control strategies; rapid antigen test.
Copyright © 2022 Barrera-Avalos, Luraschi, Vallejos-Vidal, Mella-Torres, Hernández, Figueroa, Rioseco, Valdés, Imarai, Acuña-Castillo, Reyes-López and Sandino.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
-
- Ministry of science interior G of C. Cifras Oficiales COVID-19. (2021). Available online at: https://www.gob.cl/coronavirus/cifrasoficiales/ (accessed September 16, 2021).
-
- Centers for disease control prevention. Interim Guidance for Antigen Testing for SARS-CoV-2. (2021). Available online at: https://www.cdc.gov/coronavirus/2019-ncov/lab/resources/antigen-tests-gu... (accessed September 16, 2021).
Publication types
MeSH terms
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

