Sex Hormones, Growth Hormone, and the Cornea
- PMID: 35053340
- PMCID: PMC8773647
- DOI: 10.3390/cells11020224
Sex Hormones, Growth Hormone, and the Cornea
Abstract
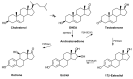
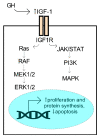
The growth and maintenance of nearly every tissue in the body is influenced by systemic hormones during embryonic development through puberty and into adulthood. Of the ~130 different hormones expressed in the human body, steroid hormones and peptide hormones are highly abundant in circulation and are known to regulate anabolic processes and wound healing in a tissue-dependent manner. Of interest, differential levels of sex hormones have been associated with ocular pathologies, including dry eye disease and keratoconus. In this review, we discuss key studies that have revealed a role for androgens and estrogens in the cornea with focus on ocular surface homeostasis, wound healing, and stromal thickness. We also review studies of human growth hormone and insulin growth factor-1 in influencing ocular growth and epithelial regeneration. While it is unclear if endogenous hormones contribute to differential corneal wound healing in common animal models, the abundance of evidence suggests that systemic hormone levels, as a function of age, should be considered as an experimental variable in studies of corneal health and disease.
Keywords: cornea; estradiol; estrogen; growth hormone; insulin growth factor-1; wound healing.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

