Kidney Damage Caused by Obesity and Its Feasible Treatment Drugs
- PMID: 35054932
- PMCID: PMC8775419
- DOI: 10.3390/ijms23020747
Kidney Damage Caused by Obesity and Its Feasible Treatment Drugs
Abstract
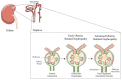
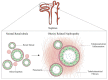
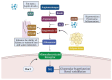
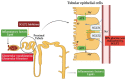
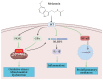
The rapid growth of obesity worldwide has made it a major health problem, while the dramatic increase in the prevalence of obesity has had a significant impact on the magnitude of chronic kidney disease (CKD), especially in developing countries. A vast amount of researchers have reported a strong relationship between obesity and chronic kidney disease, and obesity can serve as an independent risk factor for kidney disease. The histological changes of kidneys in obesity-induced renal injury include glomerular or tubular hypertrophy, focal segmental glomerulosclerosis or bulbous sclerosis. Furthermore, inflammation, renal hemodynamic changes, insulin resistance and lipid metabolism disorders are all involved in the development and progression of obesity-induced nephropathy. However, there is no targeted treatment for obesity-related kidney disease. In this review, RAS inhibitors, SGLT2 inhibitors and melatonin would be presented to treat obesity-induced kidney injury. Furthermore, we concluded that melatonin can protect the kidney damage caused by obesity by inhibiting inflammation and oxidative stress, revealing its therapeutic potential.
Keywords: inflammation; kidney damage; melatonin; obesity; oxidative stress; treatment.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

