Usefulness of Cochrane Reviews in Clinical Guideline Development-A Survey of 585 Recommendations
- PMID: 35055507
- PMCID: PMC8775999
- DOI: 10.3390/ijerph19020685
Usefulness of Cochrane Reviews in Clinical Guideline Development-A Survey of 585 Recommendations
Abstract
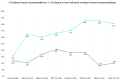
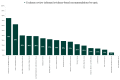
The Danish Health Authority develops clinical practice guidelines to support clinical decision-making based on the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) system and prioritizes using Cochrane reviews. The objective of this study was to explore the usefulness of Cochrane reviews as a source of evidence in the development of clinical recommendations. Evidence-based recommendations in guidelines published by the Danish Health Authority between 2014 and 2021 were reviewed. For each recommendation, it was noted if and how Cochrane reviews were utilized. In total, 374 evidence-based recommendations and 211 expert consensus recommendations were published between 2014 and 2021. Of the 374 evidence-based recommendations, 106 included evidence from Cochrane reviews. In 28 recommendations, all critical and important outcomes included evidence from Cochrane reviews. In 36 recommendations, a minimum of all critical outcomes included evidence from Cochrane reviews, but not all important outcomes. In 33 recommendations, some but not all critical outcomes included evidence from Cochrane reviews. Finally, in nine recommendations, some of the important outcomes included evidence from Cochrane reviews. In almost one-third of the evidence-based recommendations, Cochrane reviews were used to inform clinical recommendations. This evaluation should inform future evaluations of Cochrane review uptake in clinical practice guidelines concerning outcomes important for clinical decision-making.
Keywords: Cochrane; GRADE; clinical practice guideline; core outcome set; systematic review.
Conflict of interest statement
All authors hold positions within The Danish Health Authority. After submitting the present paper, C.B.K. has acquired a position as research assistant with Cochrane Denmark. H.K.A. previously (until 2018) held a position as Managing Editor for the Cochrane Colorectal Group. The authors declare no other conflicts of interest.
Figures
Similar articles
-
The future of Cochrane Neonatal.Early Hum Dev. 2020 Nov;150:105191. doi: 10.1016/j.earlhumdev.2020.105191. Epub 2020 Sep 12. Early Hum Dev. 2020. PMID: 33036834
-
No. 367-2019 Canadian Guideline for Physical Activity throughout Pregnancy.J Obstet Gynaecol Can. 2018 Nov;40(11):1528-1537. doi: 10.1016/j.jogc.2018.07.001. Epub 2018 Oct 5. J Obstet Gynaecol Can. 2018. PMID: 30297272
-
The Effectiveness of Integrated Care Pathways for Adults and Children in Health Care Settings: A Systematic Review.JBI Libr Syst Rev. 2009;7(3):80-129. doi: 10.11124/01938924-200907030-00001. JBI Libr Syst Rev. 2009. PMID: 27820426
-
Nonpharmacological Treatment of Persistent Postconcussion Symptoms in Adults: A Systematic Review and Meta-analysis and Guideline Recommendation.JAMA Netw Open. 2021 Nov 1;4(11):e2132221. doi: 10.1001/jamanetworkopen.2021.32221. JAMA Netw Open. 2021. PMID: 34751759 Free PMC article.
-
Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome.Fertil Steril. 2018 Aug;110(3):364-379. doi: 10.1016/j.fertnstert.2018.05.004. Epub 2018 Jul 19. Fertil Steril. 2018. PMID: 30033227 Free PMC article. Review.
Cited by
-
Availability of evidence and comparative effectiveness for surgical versus drug interventions: an overview of systematic reviews and meta-analyses.BMJ Open. 2024 Jan 9;14(1):e076675. doi: 10.1136/bmjopen-2023-076675. BMJ Open. 2024. PMID: 38195174 Free PMC article.
-
Reliability and reproducibility of systematic reviews informing the 2020-2025 Dietary Guidelines for Americans: a pilot study.Am J Clin Nutr. 2025 Jan;121(1):111-124. doi: 10.1016/j.ajcnut.2024.10.013. Epub 2024 Dec 12. Am J Clin Nutr. 2025. PMID: 39755432 Free PMC article.
-
Metacognitive training (MCT) for psychosis: a systematic review and grade recommendations.Eur Psychiatry. 2025 May 26;68(1):e80. doi: 10.1192/j.eurpsy.2025.10027. Eur Psychiatry. 2025. PMID: 40415540 Free PMC article.
-
Availability of evidence and comparative effectiveness for surgical versus drug interventions: an overview of systematic reviews.medRxiv [Preprint]. 2023 Feb 1:2023.01.30.23285207. doi: 10.1101/2023.01.30.23285207. medRxiv. 2023. Update in: BMJ Open. 2024 Jan 9;14(1):e076675. doi: 10.1136/bmjopen-2023-076675. PMID: 36778340 Free PMC article. Updated. Preprint.
-
Mechanical ventilation core outcome set uptake in Cochrane systematic reviews: A cross-sectional study.Cochrane Evid Synth Methods. 2024 Jan 10;2(1):e12038. doi: 10.1002/cesm.12038. eCollection 2024 Jan. Cochrane Evid Synth Methods. 2024. PMID: 40476193 Free PMC article.
References
-
- Schünemann H.J., Wiercioch W., Etxeandia-Ikobaltzeta I., Falavigna M., Santesso N., Mustafa R., Ventresca M., Brignardello-Petersen R., Laisaar K.-T., Kowalski S., et al. Guidelines 2.0: Systematic development of a comprehensive checklist for a successful guideline enterprise. CMAJ Can. Med. Assoc. J. 2014;186:E123–E142. doi: 10.1503/cmaj.131237. - DOI - PMC - PubMed
-
- World Health Organization . WHO Handbook for Guideline Development. World Health Organization; Geneva, Switzerland: 2014. [(accessed on 28 December 2020)]. Available online: https://apps.who.int/iris/handle/10665/145714.
Publication types
LinkOut - more resources
Full Text Sources

