Variant-dependent heterogeneity in amyloid β burden in autosomal dominant Alzheimer's disease: cross-sectional and longitudinal analyses of an observational study
- PMID: 35065037
- PMCID: PMC8956209
- DOI: 10.1016/S1474-4422(21)00375-6
Variant-dependent heterogeneity in amyloid β burden in autosomal dominant Alzheimer's disease: cross-sectional and longitudinal analyses of an observational study
Abstract
Background: Insights gained from studying individuals with autosomal dominant Alzheimer's disease have broadly influenced mechanistic hypotheses, biomarker development, and clinical trials in both sporadic and dominantly inherited Alzheimer's disease. Although pathogenic variants causing autosomal dominant Alzheimer's disease are highly penetrant, there is substantial heterogeneity in levels of amyloid β (Aβ) between individuals. We aimed to examine whether this heterogeneity is related to disease progression and to investigate the association with mutation location within PSEN1, PSEN2, or APP.
Methods: We did cross-sectional and longitudinal analyses of data from the Dominantly Inherited Alzheimer's Network (DIAN) observational study, which enrols individuals from families affected by autosomal dominant Alzheimer's disease. 340 participants in the DIAN study who were aged 18 years or older, had a history of autosomal dominant Alzheimer's disease in their family, and who were enrolled between September, 2008, and June, 2019, were included in our analysis. 206 participants were carriers of pathogenic mutations in PSEN1, PSEN2, or APP, and 134 were non-carriers. 62 unique pathogenic variants were identified in the cohort and were grouped in two ways. First, we sorted variants in PSEN1, PSEN2, or APP by the affected protein domain. Second, we divided PSEN1 variants according to position before or after codon 200. We examined variant-dependent variability in Aβ biomarkers, specifically Pittsburgh-Compound-B PET (PiB-PET) signal, levels of CSF Aβ1-42 (Aβ42), and levels of Aβ1-40 (Aβ40).
Findings: Cortical and striatal PiB-PET signal showed striking variant-dependent variability using both grouping approaches (p<0·0001), despite similar progression on the clinical dementia rating (p>0·7), and CSF Aβ42 levels (codon-based grouping: p=0·49; domain-based grouping: p=0·095). Longitudinal PiB-PET signal also varied across codon-based groups, mirroring cross-sectional analyses.
Interpretation: Autosomal dominant Alzheimer's disease pathogenic variants showed highly differential temporal and regional patterns of PiB-PET signal, despite similar functional progression. These findings suggest that although increased PiB-PET signal is generally seen in autosomal dominant Alzheimer's disease, higher levels of PiB-PET signal at an individual level might not reflect more severe or more advanced disease. Our results have high relevance for ongoing clinical trials in autosomal dominant Alzheimer's disease, including those using Aβ PET as a surrogate marker of disease progression. Additionally, and pertinent to both sporadic and autosomal dominant Alzheimer's disease, our results suggest that CSF and PET measures of Aβ levels are not interchangeable and might reflect different Aβ-driven pathobiological processes.
Funding: National Institute on Aging, Doris Duke Charitable Foundation, German Center for Neurodegenerative Diseases, Japanese Agency for Medical Research and Development.
Copyright © 2022 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of interests JPC has served on medical advisory boards for Otsuka Pharmaceuticals and Humana Healthcare, outside of the submitted work. APS has served on medical advisory boards for Janssen Pharmaceuticals and Biogen, outside of the submitted work. SMS reports consulting to Eisai, Novartis, Genentech, F Hoffmann-La Roche, Gemvax, Avid Radiopharmaceuticals, and Eli Lilly, outside of the submitted work. SMS also serves on steering committees for major biomarker and clinical trials and consortia such as the Alzheimer's Disease Neuroimaging Initiative, the Dominantly Inherited Alzheimer Network (DIAN), Alzheimer's Clinical Trial Consortium, Global Alzheimer's Platform- Network, and Longitudinal Early-onset Alzheimer's disease study, and is a project arm leader for the Dominantly Inherited Alzheimer's Network-Treatment Unit (DIAN-TU) study, outside of the submitted work. WEK is a co-inventor of Pittsburgh-Compound-B (PiB) and, as such, has a financial interest in a license agreement held by GE Healthcare and the University of Pittsburgh (PA, USA) based on the PiB-PET technology used in this manuscript. GE Healthcare provided no grant support for this study and had no role in the design or interpretation of results or preparation of this manuscript. AMF has received research funding from the National Institutes of Health and National Institute on Aging, Biogen, Centene, Fujirebio, and Roche Diagnostics, outside of the submitted work. AMF is a member of the scientific advisory boards for Roche Diagnostics, Genentech, and AbbVie, and also consults for Araclon and Grifols, DiademRes, DiamiR, and Otsuka Pharmaceuticals, outside of the submitted work. RJB has equity ownership interest in C2N Diagnostics and receives royalty income based on technology (ie, stable isotope labelling kinetics and a blood plasma assay) licensed by Washington University (WA, USA) to C2N Diagnostics. RJB receives income from C2N Diagnostics for serving on the scientific advisory board. Washington University, with RJB as co-inventor, has submitted the US non-provisional patent application “Cerebrospinal fluid (CSF) tau rate of phosphorylation measurement to define stages of Alzheimer's disease and monitor brain kinases/phosphatases activity.” RJB has received honoraria from Janssen and Pfizer as a speaker, and from Merck and Pfizer as an advisory board member. RJB has been an invited speaker, advisory board member, and consultant for F Hoffman La Roche, an invited speaker and consultant for AC Immune and Janssen, and a consultant for Amgen and Eisai, outside of the submitted work. AMG has consulted for Eisai, Biogen, Pfizer, AbbVie, Cognition Therapeutics, and GSK, outside of the submitted work. AMG also served on the Scientific Advisory Board of Denali Therapeutics (from 2015 to 2018), outside of the submitted work. RAS and KAJ are involved in public-private partnership clinical trials sponsored by the National Institutes of Health and Eli Lilly, which owns the distribution rights to flortaucipir, but they do not have any personal financial relationship with Eli Lilly. NG-R reports grants from Biogen, Abbvie, and Lilly, outside of the submitted work. All other authors declare no competing interests.
Figures


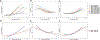

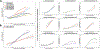
Comment in
-
From patients to disease: the difficult case of Alzheimer's.Lancet Neurol. 2022 Feb;21(2):105-106. doi: 10.1016/S1474-4422(21)00461-0. Lancet Neurol. 2022. PMID: 35065027 No abstract available.
References
-
- Borchelt DR, Thinakaran G, Eckman CB, et al. Familial Alzheimer’s Disease–Linked Presenilin 1 Variants Elevate Aβ1–42/1–40 Ratio In Vitro and In Vivo. Neuron 1996; 17(5): 1005–13. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

