Identifying the clinical presentations, progression, and sequela of pelvic inflammatory disease through physiological, histological and ultrastructural evaluation of a rat animal model
- PMID: 35071404
- PMCID: PMC8743706
- DOI: 10.21037/atm-21-3345
Identifying the clinical presentations, progression, and sequela of pelvic inflammatory disease through physiological, histological and ultrastructural evaluation of a rat animal model
Abstract
Background: Patients with pelvic inflammatory disease (PID) are at an increased risk of ectopic pregnancy, infertility, and varying degrees of chronic pelvic pain. The aims of this study were to establish a rat model of PID and characterize its progression in order to assist in the study of pathophysiological mechanisms and to provide animal model for future studies of PID treatments.
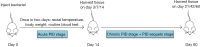
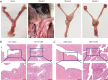
Methods: Fifty Sprague-Dawley rats (female, 6-weeks-old) were divided into a model group (n=28) and a control group (n=22). The rat endometrium was mechanically injured by a needle which moved back and forth 3 times on the endometrial tissue, and a mixed bacterial solution (6×108 CFU) of equal concentrations of Escherichia coli and Staphylococcus aureus was injected into both horns of the rat uterus. Physiological characteristics including weight, temperature, blood, and inflammatory factors were compared, and immunohistochemistry and transmission electron microscopy were used to evaluate the progress and sequela of PID.
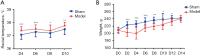
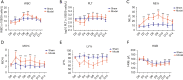
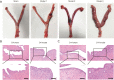
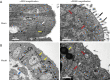
Results: The model rats experienced acute PID in the first 14 days and exhibited higher body temperatures and decreased body weight. Infection-related factors in the blood were also significantly changed compared with the normal group, with obviously increased serum levels of C-reactive protein (CRP), interferon gamma (IFN-γ), and interleukin-4 (IL-4). Congestion and edema were observed in the uteri of the model rats, followed by infiltration of numerous inflammatory cells and ultrastructural morphology changes. Histological examination of the uterus showed that adhesion initially appeared at approximately 21 days. In addition to the increased collagen fibers biomass, the expression of transforming growth factor-beta 1 (TGF-β1) was elevated, which might have contributed to pelvic tissue adhesion formation in the PID sequela.
Conclusions: This study clearly described the characteristics and progression of PID in a rat model. The detailed evidence increased our understanding of the pathogenesis and progression of PID and may be useful for future studies of PID treatments.
Keywords: Pelvic inflammatory disease (PID); rat model; tissue adhesion; transforming growth factor-beta 1 (TGF-β1).
2021 Annals of Translational Medicine. All rights reserved.
Conflict of interest statement
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://dx.doi.org/10.21037/atm-21-3345). The authors have no conflicts of interest to declare.
Figures
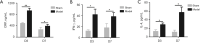

Similar articles
-
Kangfuxiaoyanshuan alleviates uterine inflammation and adhesion via inhibiting NF-κB p65 and TGF-β/MMP-2 signaling pathway in pelvic inflammatory disease rats.Front Pharmacol. 2022 Jul 18;13:894149. doi: 10.3389/fphar.2022.894149. eCollection 2022. Front Pharmacol. 2022. PMID: 35924054 Free PMC article.
-
Punicalagin improves inflammation and oxidative stress in rat model of pelvic inflammatory disease.Nat Prod Res. 2025 May;39(10):2780-2786. doi: 10.1080/14786419.2024.2313183. Epub 2024 Feb 8. Nat Prod Res. 2025. PMID: 38329023
-
Ozone therapy ameliorates inflammation and endometrial injury in rats with pelvic inflammatory disease.Biomed Pharmacother. 2018 Nov;107:1418-1425. doi: 10.1016/j.biopha.2018.07.137. Epub 2018 Sep 1. Biomed Pharmacother. 2018. PMID: 30257358
-
Acute pelvic inflammatory disease.Urol Clin North Am. 1984 Feb;11(1):65-81. Urol Clin North Am. 1984. PMID: 6369707 Review.
-
Incidence, prevalence, and trends of acute pelvic inflammatory disease and its consequences in industrialized countries.Am J Obstet Gynecol. 1980 Dec 1;138(7 Pt 2):880-92. doi: 10.1016/0002-9378(80)91077-7. Am J Obstet Gynecol. 1980. PMID: 7008604 Review.
Cited by
-
Exploring the therapeutic effect of Pen Yan Kang Fu Decoction on SPID rats based on LIF/JAK2/STAT3 signaling pathway.3 Biotech. 2024 May;14(5):134. doi: 10.1007/s13205-024-03981-0. Epub 2024 Apr 23. 3 Biotech. 2024. PMID: 38665879 Free PMC article.
-
Kangfuxiaoyanshuan alleviates uterine inflammation and adhesion via inhibiting NF-κB p65 and TGF-β/MMP-2 signaling pathway in pelvic inflammatory disease rats.Front Pharmacol. 2022 Jul 18;13:894149. doi: 10.3389/fphar.2022.894149. eCollection 2022. Front Pharmacol. 2022. PMID: 35924054 Free PMC article.
-
Modified Hongteng Baijiang decoction enema improves sequelae of pelvic inflammatory disease by regulating the LIF/JAK2/STAT3 pathway and gut microbiota.Immun Inflamm Dis. 2024 Jun;12(6):e1300. doi: 10.1002/iid3.1300. Immun Inflamm Dis. 2024. PMID: 38896093 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
